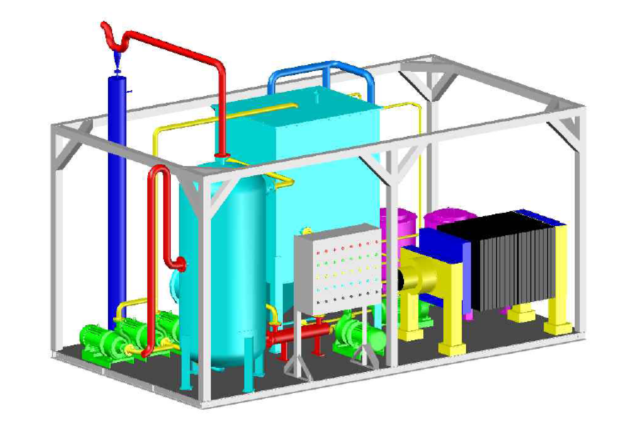
I. Technical Principle
Hydrodesulfurization using complexed iron is a liquid-phase redox technology that uses complexed iron solvent as a catalyst to directly oxidize hydrogen sulfide gas into sulfur.
Chemical Reaction:
Absorption Reaction: H2S + 2Fe3+ → 2H+ + S↓ + 2Fe2+
★Regeneration Reaction: 2Fe2+ + 1/2O2 + H2O → 2Fe3+ + 2OH-
★Overall Reaction: H2S + 1/2O2 → S↓ + H2O
Section II: Technical Advantages
This technology boasts high desulfurization efficiency, rapid oxidation and regeneration, easy recovery of sulfur, and non-polluting desulfurizing agents. The process is simple, operates at room temperature and low pressure, requires no external heat source, has low energy consumption, and offers a wide operational flexibility, making it suitable for treating gases with any hydrogen sulfide content.
★ Comprehensive biogas purification and desulfurization in one step, no need for additional dry purification.
★ Skid-mounted equipment, short process flow, compact structure, small land occupation
★ Investment and operating costs are both lower than traditional wet flue gas desulfurization and biological desulfurization.
★ By-products include elemental sulfur; no waste liquid is discharged during the production process, ensuring no pollution.
Application Fields:
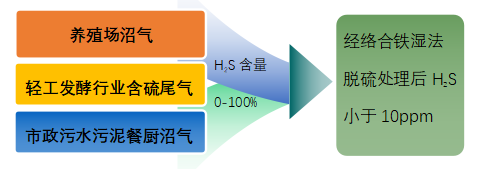
Section 3: Process Flow
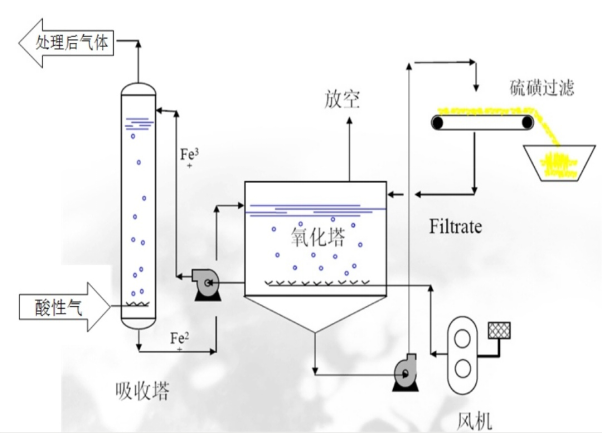
Hydrogen sulfide-containing acidic gases enter the absorption tower, where they undergo a thorough mixing and reaction with the solution to remove the hydrogen sulfide. The trivalent iron ions in the solution react with hydrogen sulfide and convert to bivalent iron ions.
A solution rich in ferrous ions is pumped into the oxidation tower, where it reacts with air forced in by the fan. The ferrous ions in the rich liquid are oxidized by oxygen to become ferric ions in the lean liquid, which is then recirculated in the absorption tower by a lean liquid pump.
The elemental sulfur settles to the bottom of the oxidation tower, and once the sulfur content reaches a certain level, the valve is opened to feed it into the filter separator to isolate the elemental sulfur.












































