Primary Uses: Biopharmaceuticals, in vitro diagnostic reagents, medical devices, pharmaceutical industry, oral liquids, cosmetics, food, large-volume parenterals, pharmaceutical preparations, genetic engineering, renal dialysis, etc., as well as water, disinfectants, non-woven fabric industry, medical and health supplies (gauze, masks, wet wipes, protective clothing, medical gloves, drainage tubes, etc.)

※Pure Environmental Protection Brand『Design Advantages』:
1. One-on-one professional service, free on-site site survey, considering placement area and entry space, free original water quality testing.
2. Understand the client's full-day water demand, peak water usage, water quality requirements, and their production process and industry characteristics.
3. Professional team collaboration in design, combined with on-site inspections by dedicated service personnel, to offer a one-stop water treatment solution service.
4. Intelligent design with precise control and high level of automation. Units can perform automatic forward/reverse washing and operation, with interlock protection and alarm indication. Features 4-20mA data openness, allowing remote transmission to the DCS control system, truly achieving unattended operation.
5. Segmentally inspect according to process requirements, capable of testing parameters such as conductivity, resistivity, pH, ORP, temperature, flow rate, velocity, and pressure, ensuring that the water in each process stage meets quality standards and does not contaminate the finished product water downstream, emphasizing process control.
6. Design products tailored to the client's needs, balancing the principles of comprehensive investment and operational costs.
※Chinese Pure Environmental Protection Brand 『Production Advantages』:
1. Core components are all imported, and we have established long-term cooperative relationships with numerous international brands such as the U.S. DOW, Hydranet, GE, Siemens, Rhone-Poulenc, Grundfos of Denmark, Rosemount, Nippon Paint of Japan, and Schneider, ensuring reliable product quality.
2. Suppliers conduct product evaluations, eliminating suppliers with poor quality and service, and selecting the best to be enrolled in the qualified supplier system.
3. Strictly adhere to the quality management system; quality is paramount. Conduct incoming material appearance and performance inspections, production process inspections, and final product quality checks.
4. Production staff produce according to assigned numbers, organized in shifts for production, with responsibility-based division of labor. Establish a performance assessment system with strong traceability.
5. Pre-shipment trial operation inspection and electrical point operation test to ensure delivery of qualified products to the customer.
※Pure Eco-Friendly Brand - "After-Sales Advantages":
1. Professional installation and after-sales team offering one-on-one service, enhancing the timeliness of post-installation after-sales support and boosting customer satisfaction.
2. Establish a post-installation after-sales service system, conduct performance evaluations, set up a customer service department, a complaint window, and promptly address customer concerns.
3. Full guidance and training throughout the installation and debugging process, including theoretical training, hands-on machine operation training, and general troubleshooting training.
4. Establish customer electronic profiles, conduct monthly phone follow-ups, and promptly provide methods and suggestions to reduce operation costs for equipment.
5. Multi-channel after-sales feedback, offering nationwide after-sales maintenance services, with quick response and rapid handling, 24/7 service.
6. Strictly Commit: Two-year free warranty for the entire unit, lifetime free technical support.

I. GMP Pharmaceutical Purified Water Production Process:
1. Grade 2 RO: Conductivity of water ≤ 2 us/cm (25℃), all other specifications comply with current pharmacopoeia GMP/FDA purified water standards.
Raw Water → Raw Water Tank → Boost Pump → Sand Filter → Carbon Filter → Softener → Security Filter → High-Pressure Pump → First Stage Reverse Osmosis → First Stage Tank → PH Adjustment → High-Pressure Pump → Second Stage Reverse Osmosis → Purified Water Tank → UV Sterilizer → Pure Water Transfer Pump → Fine Filter → Water Usage Point → Return to Purified Water Tank
Note: Utilize point-of-loop sterilization methods: Ozone sterilization/Pasteurization/Hydrogen peroxide sterilization
2. Grade 1 RO+EDI: Water conductivity ≤ 0.2 us/cm (25°C), all other specifications comply with the current pharmacopeia GMP/FDA purified water standards.
Source Water → Source Water Tank → Boost Pump → Sand Filter → Carbon Filter → Softener → Security Filter → High-Pressure Pump → First Stage Reverse Osmosis → First Stage Water Tank → Boost Pump → Fine Filter → EDI Desalination Unit → Purified Water Tank → UV Sterilizer → Pure Water Transfer Pump → Fine Filter → Water Usage Point → Return to Purified Water Tank
Note: 1. Utilize point-of-loop sterilization methods: Ozone sterilization/Batch sterilization/Hydrogen peroxide sterilization
2. This process is suitable for areas with good source water.
3. Grade 2 RO+EDI: Product water resistivity ≥15 MΩ·cm (25°C), all other specifications comply with current pharmacopoeia GMP/FDA purified water standards.
Source Water → Source Water Tank → Booster Pump → Sand Filter → Carbon Filter → Softener → Security Filter → High-Pressure Pump → Primary Reverse Osmosis → Primary Water Tank → PH Adjustment → High-Pressure Pump → Secondary Reverse Osmosis → Secondary Water Tank → Booster Pump → Fine Filter → EDI Desalination Unit → Purified Water Tank → UV Sterilizer → Pure Water Delivery Pump → Fine Filter → Water Usage Point → Return to Purified Water Tank
Note: 1. Sterilization using point-of-loop pipelines: Ozone sterilization / Pasteurization / Hydrogen peroxide sterilization
2. Recommend using the process
4. Grade 1 RO + Grade 3 mixed: Water production resistivity ≥ 5 MΩ·cm (25℃), all other specifications meet the current Pharmacopoeia GMP/FDA purified water standards.
Raw Water → Activated Carbon Filter Core → 10um Filter Core → 5um Filter Core → High Pressure Pump → Reverse Osmosis RO System → First Grade Polishing Mixed Bed → Second Grade Polishing Mixed Bed → Third Grade Polishing Mixed Bed → Fine Filter → Pure Water Storage Tank → UV Sterilizer → Pure Water Delivery Pump → Fine Filter → Water Usage Point → Return to Pure Water Storage Tank
Note:
1. Sterilization via point-of-loop pipeline: Ozone sterilization / Pasteurization / Peroxide sterilization
2. Suitable for small pure water units: Flow range: 50L/H - 200L/H
For more product details, please call our 24-hour hotline at 18550863818 or our service hotline at 0512-69390898.
We will provide you with detailed plans, drawings, and competitive quotes for your reference and selection!
※ Call for consultation, enjoy 10% off! Welcome to call!
Section II: Automatic Control Description for GMP Pharmaceutical Purified Water
The original pump is controlled in series by the liquid level sensor on the raw water tank. When the liquid level in the raw water tank reaches the mid-limit, the original pump automatically starts according to the system settings. When the raw water tank is at low water level, the original pump stops delivering to prevent damage from operating without water. The original pump can only restart供水 when the liquid level in the raw water tank returns to the mid-limit.
2. Sand filtration, carbon filtration, and softening processes are all fully automated, with the flushing cycle determined through actual verification.
3. The primary high-pressure pump is controlled by the system's operational program. When the system startup pressure reaches the required value, the PLC signals, and the primary high-pressure pump automatically starts. The pump's inlet and outlet are designed with high and low-pressure protective switches. If the inlet or outlet pressure exceeds the limit, the primary high-pressure pump stops automatically according to the operational program settings to prevent damage from working under low pressure or damaging the reverse osmosis membrane under high pressure. If the primary high-pressure pump stops due to overloading of pressure, temperature, or power, the system emits an alarm according to the operational program settings. The primary high-pressure pump can only be restarted by the operator after the fault is resolved.
4. The secondary high-pressure pump is controlled by the intermediate tank's liquid level interlock; when the intermediate tank's liquid level reaches or exceeds the middle limit level, the PLC sends a signal to automatically start the secondary high-pressure pump; when the intermediate tank's liquid level falls below the low-low limit level, the PLC sends a signal to automatically stop the secondary high-pressure pump to prevent damage from operating at low pressure; the outlet of the secondary high-pressure pump is designed with a high-pressure protection device, which automatically stops the pump according to the operating program when the outlet pressure exceeds the limit, to prevent damage to the reverse osmosis membrane under high pressure; when the secondary high-pressure pump stops due to overloading of pressure, temperature, or power, the system emits an alarm according to the operating program settings; the secondary high-pressure pump can only be restarted by the operator after the fault is resolved.
5. The reverse osmosis unit is equipped with sufficient online gauges to visually reflect the unit's operating status, including: pressure gauges, flow meters, conductivity, pH, etc. A local control panel is set up for on-site operation and control.
6. The first-class reverse osmosis design features an automatic flushing function. The system automatically enters a low-pressure, high-flow flushing state every time it is powered on or runs for 4-6 hours, with a flushing duration of 15-30 seconds.
7. The EDI unit is equipped with sufficient on-line instruments that can visually reflect the unit's operating status, including pressure gauges, flowmeters, conductivity, etc., with local control panels set up for on-site operation and control. The EDI unit is interlocked with the system's operational program for control.
8. The EDI unit is designed with a low-water protection device at the water inlet, to prevent damage to the EDI unit when operated without water.
9. Continuous monitoring of the conductivity of the EDI unit's output water, interlocking control of the EDI output conductivity detection unit and the EDI output qualified and unqualified valves; automatically open the EDI output unqualified valve and close the qualified valve when the conductivity of the EDI output water exceeds the limit; automatically close the unqualified valve and open the qualified valve when the conductivity of the EDI output water returns to the set standard, to prevent unqualified water from contaminating the finished water.
10、NaOHThe addition device is controlled fully automatically, equipped with a metering pump that automatically injects diluted NaOH solution into the pipeline, ensuring precise measurement. The overall design is lightweight, with accurate speed control, and is unaffected by heat sources or electromagnetic interference. It can make appropriate adjustments based on water volume and flow rate changes through the metering pump, automatically adding chemicals. This metering pump is linked to the first-stage high-pressure pump, strictly controlling the pH value of the second-stage reverse osmosis feedwater, ensuring stable water quality of the second-stage RO product.
11. During system operation, when the pure water storage tank is full, the secondary RO and EDI systems continue to run continuously. The EDI product water is recycled to the intermediate water tank, achieving an internal small circulation to reduce the risk of dead water in the system and the potential for microbial growth.
12. The purified water storage tank is monitored and controlled by an internal liquid level gauge, equipped with a static pressure level controller that outputs a 4-20mA signal. The purified water storage tank is set up with 4 liquid level points:
13. The purified water delivery pump is controlled by the liquid level linkage of the purified water storage tank; when the liquid level in the storage tank reaches the mid-limit, the pump automatically starts according to the system settings; when the liquid level is at low water level, the pump stops delivering to prevent damage from operating without water; the pump will only start supplying water again when the liquid level in the storage tank returns to the mid-limit or exceeds it.
14. The return water monitoring system is set on the point-loop pipeline with a 4-20mA signal output. The return water flow rate is automatically controlled by varying the frequency of the pure water delivery pump via feedback from the return water flowmeter to ensure the return water flow rate is not less than the set flow rate value (recommended set value: 1.0-2.0 m/s).
15. Continuous monitoring of the conductivity online continuity in the pure water storage and distribution system's return water. Interlock control of the return water conductivity detection device, the return water qualified valve, and the return water discharge valve. When the detected return water conductivity exceeds the limit, the return water qualified valve is automatically closed, and the return water discharge valve is simultaneously opened for automatic discharge. When the return water conductivity returns to the set standard, the return water discharge valve is automatically closed, and the return water qualified valve is opened.
III. GMP Pharmaceutical Purified Water Validation Documents
1. Filter, Pipe, Valve Material Reports
2. Certificates of conformity, manuals, etc., for accessories of filters, pipes, valves, pumps, instruments, etc.
3. Certificate of Conformity, Instruction Manual
4. Filter Core Integrity Test Report
5. Filter, pipeline pressure test record report
6. Pipeline Pickling and Passivation Report
7. Automatic Welding Records
8. Provide endoscopic images: 20% of automatic welding points, 99.999% of manual welding points
9. Instrument Calibration Report and Certificate
10. Third-party Water Quality Monitoring Report
11. Provide DQ, FAT, SAT, IQ, PQ, OQ verification documents for the user's equipment.
IV. Introduction to Core Components of GMP Pharmaceutical Purified Water System
1. Reverse Osmosis (RO) System
Reverse osmosis (RO) membranes, also known as hyper-filtration, were invented by Lob and Sourirtajan of the University of California, Los Angeles in 1960. These advanced membrane separation technologies have pores as small as nanometers (1 nanometer = 10^-9 meters). Under certain pressure, H2O molecules can pass through the RO membrane, while inorganic salts, heavy metal ions, organic matter, colloids, bacteria, and viruses in the source water cannot pass through. This effectively separates pure water that can pass through from concentrated water that cannot.
Principle: Osmosis is a physical phenomenon where, when two bodies of water with different salt concentrations are separated by a semi-permeable membrane, the water on the side with lower salt content will pass through the membrane until the concentrations are equalized. However, this process takes a long time and is known as natural osmosis. But if a pressure is applied to the side with higher salt content, it can stop the osmosis, and this pressure is called osmotic pressure. If the pressure is increased further, it can cause the water to permeate in the opposite direction, leaving the salt behind. Therefore, the principle of reverse osmosis desalination is to apply a pressure greater than the natural osmotic pressure to saltwater (such as raw water), causing the osmosis to occur in the opposite direction, forcing the water molecules to the other side of the membrane, resulting in clean water. This is how reverse osmosis desalination removes salt from water.
The reverse osmosis membrane utilizes an aromatic polyamide spiral wound composite membrane. The reverse osmosis unit consists of composite membrane elements, high-pressure pumps, glass steel pressure vessels, chemical cleaning units, supports, and instrument control cabinets. Equipped with a local control panel, which houses various local instruments and control buttons. The reverse osmosis unit is controlled by a PLC for fully automatic operation. It is also equipped with flowmeters, pressure gauges, conductivity meters, and more.

2. EDI Desalination System
EDI equipment for continuous electrical deionization (EDI, Electro deionization) employs mixed ion exchange resins to absorb positive and negative ions from the feed water, while these absorbed ions are then removed by passing through anion and cation exchange membranes under the influence of direct current voltage. In this process, the ion exchange resins are electrochemically regenerated continuously, eliminating the need for acid and alkali regeneration. This new technology can replace traditional ion exchange (DI) units, producing ultra-pure water with resistivity ≥ 15 MΩ·cm.
Ion exchange membranes and ion exchange resins operate on similar principles, selectively allowing ions to pass through. A cation exchange membrane only permits cations to pass through, not anions; conversely, an anion exchange membrane allows only anions to pass, blocking cations. Filling the space between a pair of anion and cation exchange membranes with mixed ion exchange resins creates an EDI unit. The space occupied by mixed ion exchange resins between the anion and cation exchange membranes is known as the freshwater chamber. Arranging a certain number of EDI units in a row, alternating anion and cation exchange membranes, and adding special ion exchange resins between the membranes, forms a concentrate chamber. Under the influence of a given DC voltage, in the freshwater chamber, anions and cations in the ion exchange resins migrate towards the positive and negative poles, respectively, and pass through the anion and cation exchange membranes into the concentrate chamber, while ions in the feedwater are adsorbed by the ion exchange resins, occupying the voids left by ion electro-migration. In fact, ion migration and adsorption occur simultaneously and continuously. Through this process, ions in the feedwater pass through the ion exchange membranes into the concentrate chamber and are removed to become desalinated water. Negatively charged anions (e.g., OH-, Cl-) are attracted to the positive pole (+) and pass through the anion exchange membrane into the adjacent concentrate chamber. Subsequently, these ions encounter the adjacent cation exchange membrane during their continued migration towards the positive pole, which does not allow anions to pass, thus blocking these ions in the concentrate. In a similar manner, cations in the freshwater flow (e.g., Na+, H+) are blocked in the concentrate chamber. In the concentrate chamber, ions passing through the anion and cation membranes maintain electrical neutrality. The electrical current and ion migration in the EDI component are proportional. The electrical current consists of two parts: one from the migration of the removed ions, and the other from the migration of H+ and OH- ions produced by the electrical ionization of water. A high voltage gradient exists within the EDI component, causing water to electrolyze and produce a large amount of H+ and OH-. These locally produced H+ and OH- continuously regenerate the ion exchange resins. The ion exchange resins in the EDI component can be divided into two parts: the working resin and the polishing resin, with the boundary between them being the working front. The working resin is responsible for removing most of the ions, while the polishing resin is responsible for removing difficult-to-remove ions such as weak electrolytes.
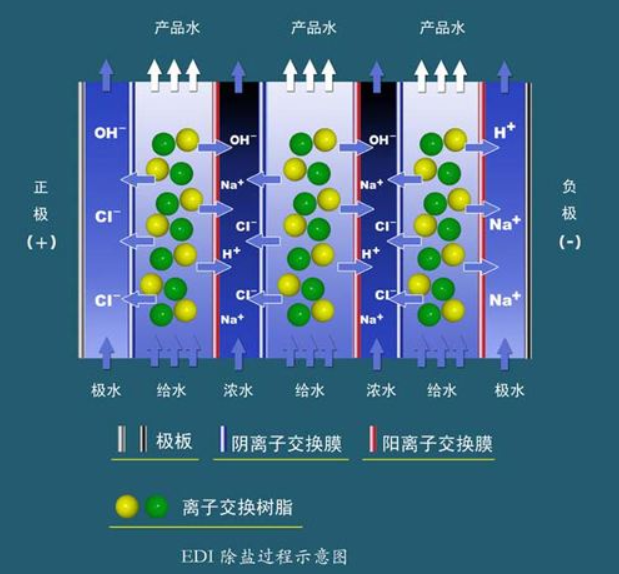
Features of EDI
1. No need for acid-base regeneration, no need for acid-base reserve and dilution transportation facilities, safe and reliable use, avoids worker exposure to acids and bases
2. Saves on recycled water and wastewater treatment facilities.
3. Reduced operational and maintenance costs
4. Compact footprint, easy installation, and high water yield (up to 90-95%)
5. Continuous operation, stable product and water quality, will not shut down due to regeneration
3. Pharmaceutical Water Supply
1) Purified water and pharmaceutical water should be transported using stainless steel pumps that are easy to disassemble for cleaning and disinfection. In situations where compressed air or nitrogen gas is required for the transport of purified water or water for injection, the compressed air and nitrogen must be purified.
2) Pure water should be transported through a recirculating pipeline. The pipeline design should be simple, avoiding blind pipes and dead ends, and must meet the 3D standards required by GMP. Threaded connections are not permitted. The pipeline should be made of stainless steel or other materials that are verified to be non-toxic, corrosion-resistant, and do not leak pollutants. Valves should be of a clean-grade design with no dead corners (recommend using clean-grade diaphragm valves), and the direction of water flow should be indicated during pure water transportation.
3) Pipes and pumps for transporting purified and injection water should be regularly cleaned, disinfected, and sterilized. They can only be put into use after verification of their compliance.
8. Design of pressure vessels must be undertaken by licensed entities and qualified personnel, in accordance with the relevant provisions of the People's Republic of China's National Standard for Steel Pressure Vessels (GB150-80) and the "Safety Supervision Regulations for Pressure Vessel Technology."
Section 5: Standard for Pure Water for Pharmaceutical Purposes/Extracellular Diagnostic Reagents Water
(1) Current edition of the Chinese Pharmacopoeia GMP requirements for purified water
Appearance: Colorless clear liquid, odorless, tasteless
2. PH:5.0~7.0
Ammonia: ≤0.3 mg/L
Conductivity: ≤1 μS/cm @ 25℃
Total Organic Carbon: ≤0.5 mg/L
6. Non-volatile matter: ≤0.1 mg/L
7. Heavy Metals: ≤0.1 mg/L
Microorganisms: ≤50 CFU/ml (Regulatory Standard: Cultured on R2A agar medium at 30~35℃ for not less than 5 days, tested in accordance with regulations (General Method 1105). The total aerobic bacteria in 1 mL of the test sample shall not exceed 100 CFU.)
(II) Standard for Water Used in In Vitro Diagnostic Reagents YY/T 1244-2014
1. This product should be colorless and transparent liquid
Conductivity: ≤1 μS/cm @ 25℃
Microorganisms: 50 cfu/ml
4. Total Organic Carbon: <500 μg/L
5. Other specifications are executed according to the purified water standards.
Section VI: Selected Classic Customer Cases:

Section 7: National Warranty and Service Network
1. Professional installation and after-sales team providing one-on-one service, enhancing the timeliness of post-installation after-sales support and improving customer satisfaction.
2. Establish a post-installation after-sales service mechanism, conduct performance evaluations, set up a customer service department, a complaint hotline, and promptly address customer needs.
3. Full guidance and training throughout the installation and debugging process, including theoretical training, hands-on machine operation training, and general troubleshooting training.
4. Establish customer electronic profiles, conduct monthly phone follow-ups, and promptly provide methods and suggestions to reduce equipment operation costs.
5. Multiple channels for after-sales feedback, nationwide after-sales maintenance services, rapid response, quick handling, 24/7 service.
6. We solemnly promise: Two-year free warranty for the entire unit, lifetime free technical support.
For more product information, please call the 24-hour hotline at 18550863818 or the service hotline at 0512-69390898.
We will provide you with detailed plans, drawings, and competitive quotes for your reference and selection!
Call for consultation, enjoy 10% off! Welcome to call!







































