Overseas Purification Engineering
Applicable Industries: Food, Electronics, GMP, Biomedicine, Medical Devices, Food & Pharmaceutical Packaging, New Energy, etc.
Suggested Grade: 100th Level - 300,000th Level












Cleanroom Construction (Cleanroom) Definition:
Cleanroom engineering involves the removal of particulates, harmful air, bacteria, and other pollutants from a specific space, as well as controlling the indoor temperature, cleanliness, pressure, air velocity and distribution, noise vibration, and lighting to meet certain requirements within a specially designed room. This means that regardless of changes in external air conditions, the room maintains the original set cleanliness, temperature and humidity, and pressure characteristics.
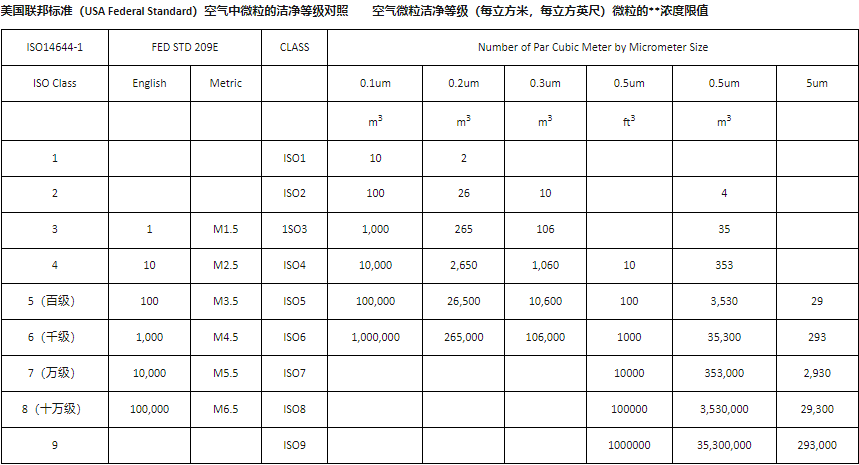
Particles smaller than 0.3 micrometers in diameter are limited to less than 3,500 per cubic meter to meet the A-grade international dust-free standard. The dust-free standards currently used in wafer-level production and processing have stricter requirements than the A-grade, with the number of microparticles strictly controlled to less than 1,000 per cubic meter—the industry's so-called 1K level.
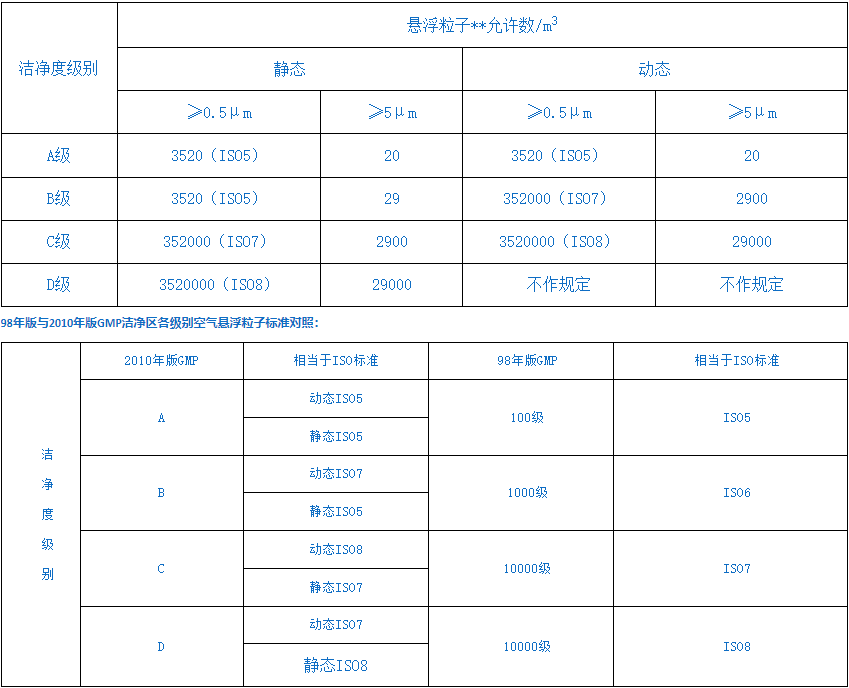
2010 Revised GMP Cleanroom Classification
Grade A: High-risk operation areas, such as filling zones, areas where stoppered barrels are placed and open-top packaging containers in direct contact with sterile preparations, as well as areas for sterile assembly or connection operations, should maintain the environmental status with unidirectional airflow benches (shelters). The unidirectional airflow system must provide uniform air supply in its working area, with a wind speed of 0.36-0.54 m/s (guideline value). Data should be available to demonstrate the unidirectional airflow status and it must be verified. Lower wind speeds can be used within enclosed isolation devices or glove boxes.
Grade B: Refers to the background area surrounding the Grade A clean zone where high-risk operations such as aseptic preparation and filling are conducted.
Class C and D: refer to clean areas in the production process of sterile pharmaceuticals that involve less critical operations.
China GMP (2010 Revision) 2010 Edition, Annex 1 of GMP, Chapter 3, Article 9...The standard specifications for air-borne particles in different grades of clean areas are as follows in the table: