Clean GMP Cleanroom
Applicable Industries: Medical Equipment, Biopharmaceuticals, Raw Material Production, Health Products, Food, Cosmetics, etc.
Suggested Grade: 100th - 300,000th Level

Clean GMP Cleanroom Construction

Clean GMP Cleanroom Construction Project

Clean GMP Manufacturing Facility Purification Project

Clean GMP Cleanroom Construction

Clean GMP Manufacturing Facility Purification Project

Clean GMP Manufacturing Facility Purification Project

Clean GMP Workshop Purification Project

Clean GMP Cleanroom Construction

Clean GMP Manufacturing Facility Purification Project
Clean Installation Project - GMP Cleanroom Technical Specifications:
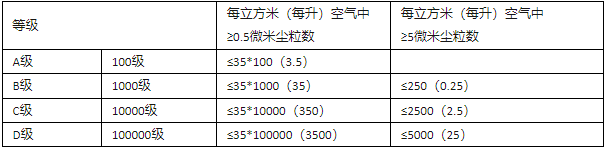
Pharmaceutical factory cleanroom areas are divided into four levels: A, B, C, and D. Industrial cleanrooms and clean zones primarily control particles and microorganisms, while also specifying requirements for environmental factors such as temperature and humidity, pressure difference, illumination, and noise. The air cleanliness of industrial clean workshops is categorized into four grades: A, B, C, and D.
Grade A Area: High-risk operation zones, including areas for filling, placing stoppered buckets, open-mouth vials, open-mouth ampoules, and sterile assembly lines or connected operations. Layered air flow benches (shelters) are typically used to maintain the environmental status of this area. The layer flow system must provide uniform air supply in its working area, with a wind speed of 0.36-0.54 m/s (guideline value). Data should be provided to demonstrate the status of the layer flow, which requires verification. In enclosed isolation operation areas or glove boxes, unidirectional flow or lower wind speeds may be used.
B-Level Area: Refers to the background area surrounding the A-Level Area where high-risk operations such as sterile preparation and filling are conducted.
C and D Class Areas: These refer to clean operation areas of lower importance in the production process of sterile pharmaceuticals.
Cleanroom Project - GMP Workshop Technical Parameters:
Old Standard: China National Standard for Design of Clean Rooms GBJ73-84
Cleanroom - GMP Cleanroom (Clean Room) Personnel Entry and Exit Regulations:
All personnel entering the clean areas of 100,000 or 300,000 class cleanrooms, including operators, maintenance staff, and support personnel, must change into clean shoes, wear cleanroom attire, and wear a clean hat; those entering 10,000 class clean areas must change into clean shoes, store their personal clothes, shower, change into cleanroom clothing, wear sterile clean attire, sanitize their hands, and pass through air showers to enter the GMP cleanroom area.
The personnel flow in the GMP cleanroom involves changing clothes and shoes in the locker room → washing hands in the sink room → the buffer room → the air shower room → various operation rooms





















.jpg)











































