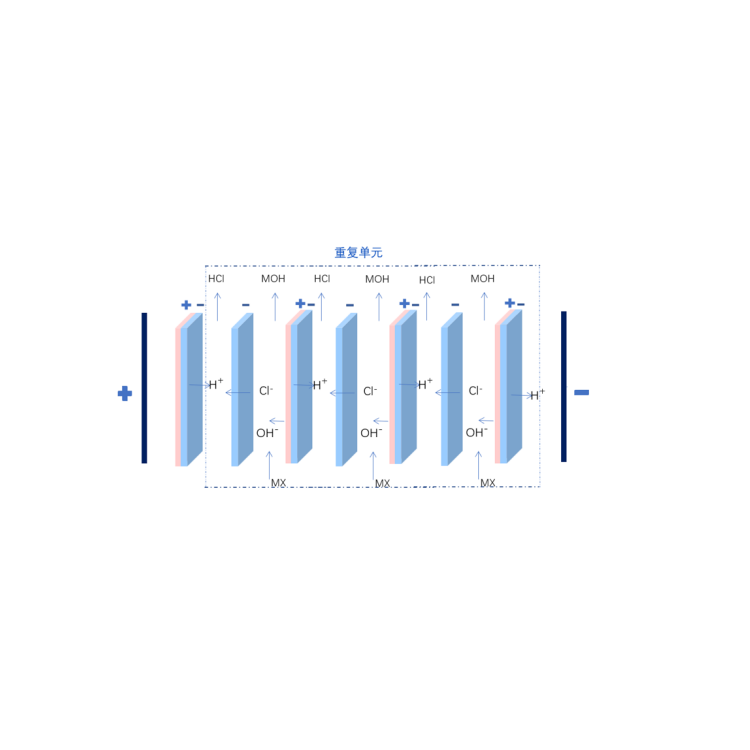
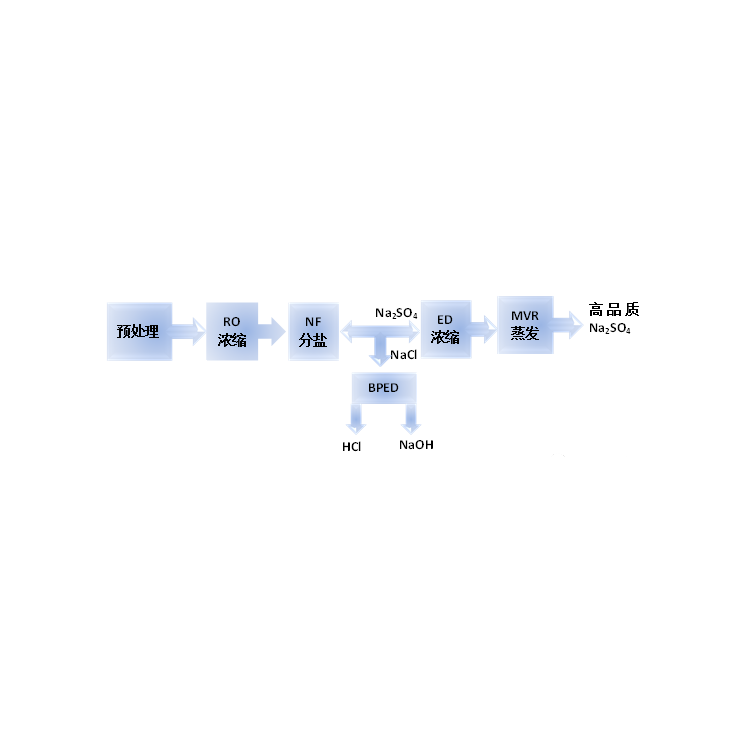
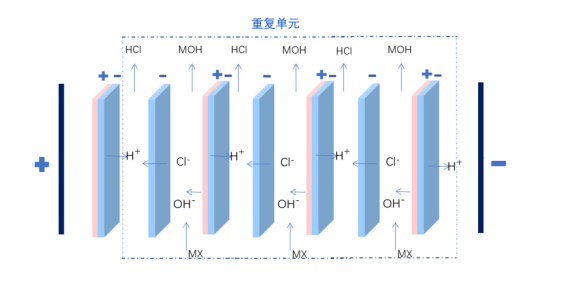
The principle of the two-compartment bipolar membrane electrodialysis lies in that when a reverse voltage is applied to both ends of the bipolar membrane, ions in the ion exchange layer of cations and anions will migrate separately through the anion and cation layers towards the bulk solution. Consequently, a narrow region with a strong electric field appears at the intermediate interface layer of the BP. At this point, water molecules rapidly dissociate into H+ and OH- ions, which migrate into the bulk solution. The water molecules consumed are replenished by diffusion from the solution outside the membrane to the intermediate interface layer. The H+ and OH- ions that migrate into the bulk solution combine with organic salts and chloride ions that pass through the anion and cation exchange membranes, forming organic bases and hydrochloric acid in the material and acid chambers.
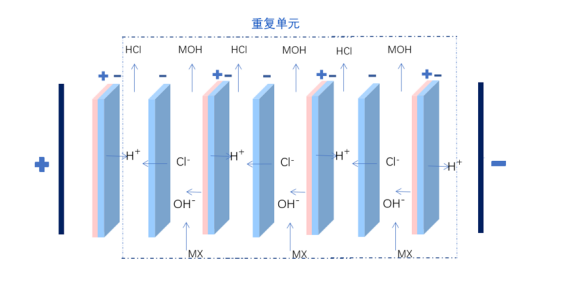
Two-compartment bipolar membrane electrodialysis for the preparation of organic bases