- AllProduct Category
-
Purification Engineering
Air Shower Room
Cleanroom Workbench
Transfer Window
Air curtain
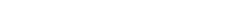

详情描述
Food Grade Sterilization and Purification Workshop
Industry Adaptability: Food, Baking, Dairy Products, Canned Goods, Seafood, Seasonings, etc.
Suggested Grades: 100th-100,000th

Food Grade Sterile Processing Facility

Food-grade无菌净化 facility

Food无菌Cleanroom

Food Sterilization and Purification Workshop

Food-grade sterile purification workshop

Food Sterilization and Purification Workshop

Food-grade Sterilization Cleanroom

Food-grade无菌净化 facility

Food无菌cleaning workshop

Food Grade Sterilization and Purification Workshop

Food Sterilization and Purification Workshop

Food-grade无菌净化 facility

Food-grade sterile purification workshop

Food-grade sterile purification workshop

Food-grade Sterile Purification Workshop

Food-grade sterile purification workshop
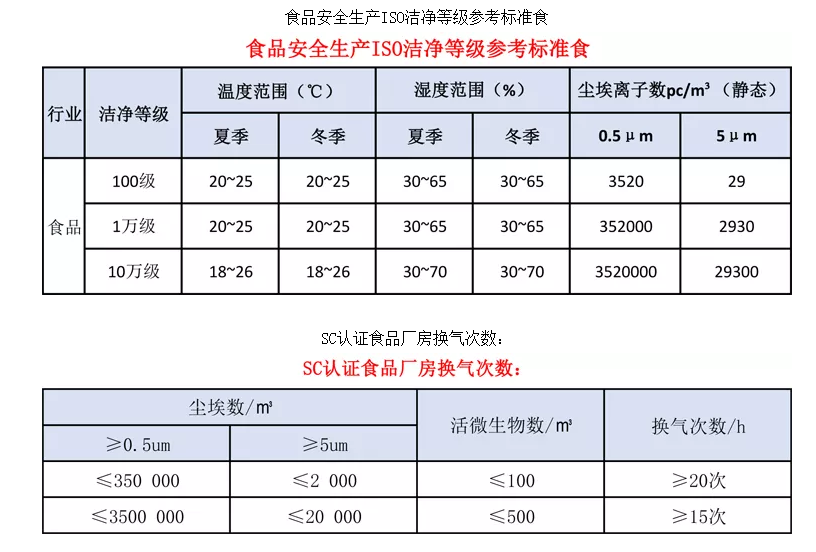
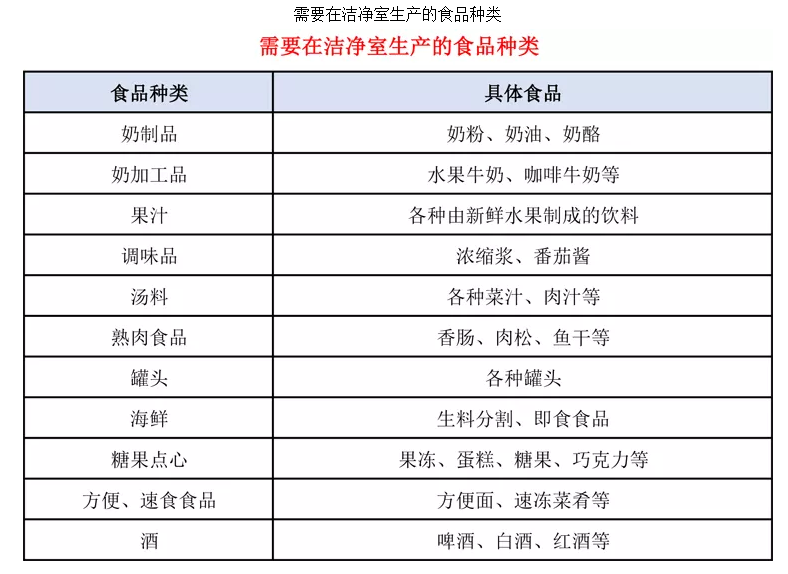
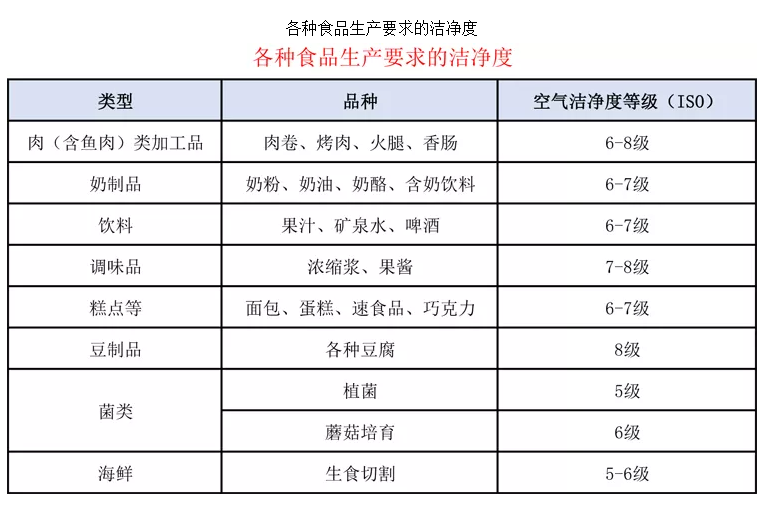
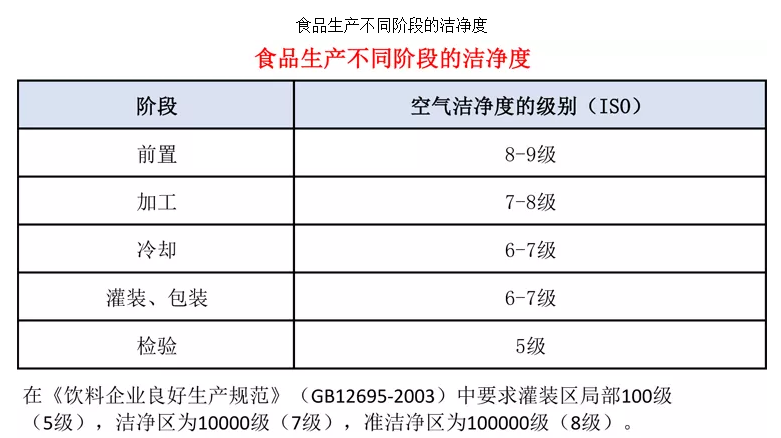
In the "Good Manufacturing Practices for Beverage Enterprises" (GB12695-2003), the filling area must meet a 100-level (Class 5) cleanliness, the clean area a 10,000-level (Class 7), and the semi-clean area a 100,000-level (Class 8) cleanliness.
Cleanroom - Air Shower for Food Grade Sterile Workshop
Cleanroom air shower rooms are an essential cleanroom accessory in modern industrial cleanrooms, capable of removing dust from the surfaces of people and objects while also acting as a buffer and isolation between the clean and non-clean zones on either side of the air shower room. This equipment is widely used in industries such as food, pharmaceuticals, biotechnology, and precision electronics. Cleanroom air shower rooms are categorized into two types: "standard" and "interlocked." The standard type uses a manual start-up blow-off control method, with some operators opting for the evasion of "leaving without passing through the blow-off." This makes it difficult to ensure the air cleanliness in the clean zone of the clean room, and it may even affect product quality.
In a sterile food processing cleanroom, under dynamic conditions, the source of bacteria and dust is the operator. Before the operator enters the aseptic room cleanroom, the surface dust particles on their clothing must be blown away with clean air.
Food无菌cleanroom operation precautions:
Sterilization and purification is achieved by filtering and removing air particles, dust, and bacteria in the food production environment, drawing from the experience in pharmaceutical production under GMP to ensure food quality.
1. The food aseptic room strictly adheres to sterile procedures to prevent microbial contamination; operators must turn off the UV light before entering the sterile room.
2. The food purification workshop should be equipped with a sterile operation room and a buffer room. The sterile operation room should reach a cleanliness level of 10,000, with indoor temperature maintained at 20-24°C and humidity at 45-60%. The clean bench should achieve a cleanliness level of 100.
3. Cleanroom sterile rooms must be kept clean, no clutter is allowed to prevent contamination. Strictly prevent any sterilization equipment and culture media from being contaminated; discontinue use if contamination occurs.
4. Cleanroom sterile rooms should be sterilized and cleaned regularly with suitable disinfectants to ensure that the cleanliness of the sterile room meets the requirements.
5. All equipment, instruments, petri dishes, and any other items required for use in a clean room sterile area, should be securely wrapped and sterilized by an appropriate method.
6. Before entering the sterile room in the clean room, staff must wash and disinfect their hands with soap or disinfectant, then change into dedicated work attire, shoes, hat, mask, and gloves (or re-wipe hands with 70% ethanol), before entering the clean room sterile room for operations.
7. Prior to use, the clean room sterile room must be sterilized by exposing the ultraviolet light for over 30 minutes, and at the same time, the clean bench should be blowing air. After operation, the sterile room should be cleaned up promptly and then sterilized again with the ultraviolet light for 20 minutes.
8. Prior to inspection, the outer packaging of the sample must remain intact and unopened to prevent contamination. Before inspection, disinfect the outer surface with a 70% alcohol swab.
9. Items containing viable bacteria must be disinfected before rinsing under the tap, and it is strictly prohibited to contaminate the drain.


Food-grade clean room decontamination workshop entry process: Changing rooms (change clothes and shoes) → Hand washing area → Buffer room → Air shower room → Various operation rooms






















.jpg)
询价单



















