- AllProduct Category
-
Purification Engineering
Air Shower Room
Cleanroom Workbench
Transfer Window
Air curtain
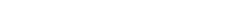

详情描述















Cleanroom Engineering (Cleanroom) Definition:
Cleanroom engineering refers to the process of removing airborne particles, harmful air, bacteria, and other pollutants within a specific space, and controlling the indoor temperature, cleanliness, air pressure, airflow speed and distribution, noise vibration, and lighting, as well as static electricity within a required range. It involves specially designed rooms that maintain the originally set cleanliness, humidity, and pressure levels regardless of external air conditions.
Each cubic meter will control the number of dust particles smaller than 0.3 micrometers to less than 3,500, achieving Grade A of international dust-free standards. The dust-free standard currently applied in wafer-level production and processing has higher requirements than Grade A, with the number of microdust particles strictly controlled to less than 1,000 per cubic meter, which is also known in the industry as the 1K level.
Cleanroom project illustration (for reference only):

Glossary (as shown in the image):
Return Air Port: Also known as the intake or exhaust port, it is the pipe opening in the middle of the air conditioning duct that transfers air to the outside.
Supply Air Diffuser: Refers to the pipe opening in the middle of the air conditioning duct that delivers air to the indoor space.
Sterile Transfer Chamber: Also known as a pass-through box, it serves as an auxiliary equipment for cleanrooms, primarily used for transferring small items between clean areas and non-clean areas to reduce the number of times the cleanroom doors are opened, effectively minimizing contamination in the clean areas.
Local Clean Rooms: Also known as clean棚, they offer various cleanliness grades and can be customized according to usage requirements. Their main features include flexibility in application, ease of installation, short construction period, and portability.
Air Shower Disinfection Zone: Also known as an air shower chamber, it is a highly versatile local purification equipment that uses a fan to blast a strong, clean air stream through nozzles, effectively removing dust particles from the surface of personnel or goods entering a clean area.
Epoxy Antistatic Flooring: Also known as epoxy floor paint, it boasts properties like resistance to strong acids and alkalis, wear and pressure resistance, impact resistance, anti-mold, waterproofing, dustproofing, skid resistance, and antistatic as well as electromagnetic wave protection. It comes in bright and varied colors and is easy to clean. The one-coat application process ensures seamless coverage regardless of the area size, and it's a dust-free material with strong adhesion, excellent wear resistance, and high hardness.
Cleanroom Bench: Also known as a clean bench, it supplements the exhausted air from the exhaust port by drawing an equivalent volume of air from the room through the operational opening. This air will never enter the operational area, serving only as an air barrier. Widely used in pharmaceutical hygiene, biopharmaceuticals, food, medical scientific experiments, optics, electronics, sterile room experiments, and sterile microbiological inspections.



010 New Edition GMP Cleanroom Classification
Grade A: High-risk operation areas, such as filling zones, areas for placing buckets with rubber stoppers, and open packaging containers directly in contact with sterile preparations, as well as areas for sterile assembly or connection operations, should maintain environmental conditions with unidirectional airflow benches (shelters). The unidirectional airflow system must provide uniform air supply within its working area at a velocity of 0.36-0.54 m/s (guideline value). Data should demonstrate the unidirectional airflow status and be verified. Lower air velocities can be used within enclosed isolators or glove boxes.
B Grade: Refers to the background area surrounding the A Grade clean zone where high-risk operations such as sterile preparation and filling take place.
C and D Grades: refer to clean areas in the production process of sterile pharmaceuticals, which involve lower importance operational steps
China GMP (2010 Revision) 2010 Edition, Annex 1 of GMP, Chapter 3, Article 9...The standard specifications for air-borne particles in different grades of clean areas are as follows in the table:








.jpg)
询价单



















