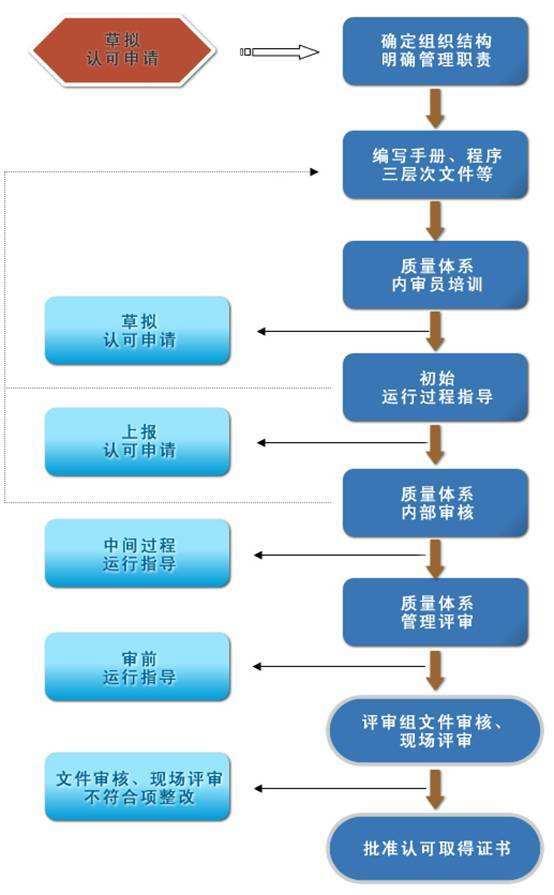
CNAS Certification Approval Service | How to Handle Various Changes for CNAS
How to handle various changes of CNAS
CNAS-RL01: 2019 9.1 Change in Laboratory Accreditation 9.1.1 Change Notification
If any of the following changes occur in an approved recognized laboratory, notice should be given to the CNAS Secretariat within 20 working days:
l The name, address, legal status, and main policies of the laboratory recognized by the approval authority have changed.
l The organization, senior management, technical staff, and authorized signatories of the approved and recognized laboratory have changed.
l Changes to the standards/methods, key testing equipment, environment, scope of testing/calibration/verification, and related projects within the recognized scope.
l Other changes that may affect business activities and system operations within its scope of recognition.
Note 1: If there are any changes to the name, address, standards/methods for testing/calibration/verification, authorized signatories, etc. of the laboratory approved for recognition, a change application must be filled out and submitted.
Note 2: Any changes to other information of the laboratory recognized by the authority (such as contact person, contact information, etc.) should be updated promptly.