Laboratory CMAI & CNAS Certification Consulting Services - Nationally Recognized Body
InspectionApplication for CNAS recognition in a laboratory requires submission of the document list.
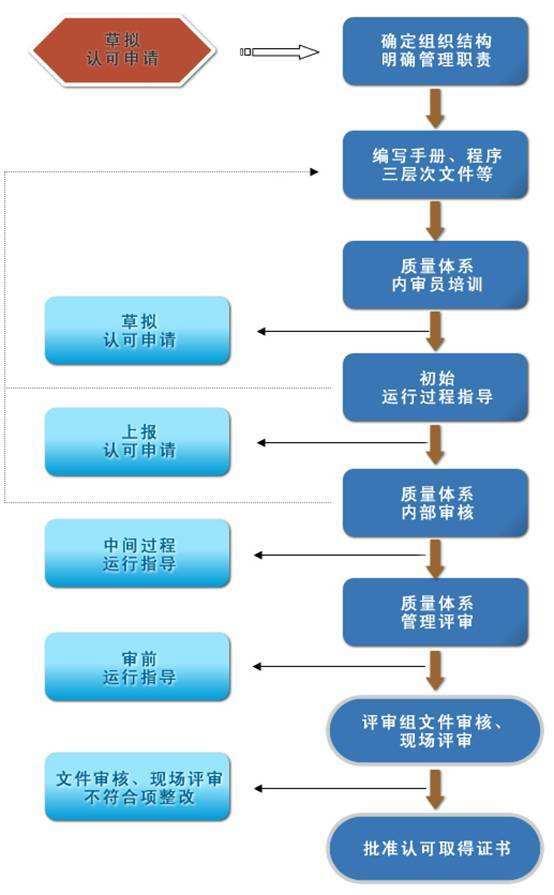
1. Acknowledgment Application
Application Form Attachment:
a) Attachment 1: Overview of Key Laboratory Locations
b) Appendix 2-1: List of Authorized Signatories for Laboratory
Appendix 2-2: Authorized Signatory Application Form
d) Attachment 3: Laboratory Staff List
e) Attachment 4-1: Application for Accreditation of Testing Capabilities and Equipment (including Standard Materials/Standard Samples) Configuration/Verification
f) Appendix 4-6: Overview of Criteria for Evaluation (Laboratories not applying for evaluation may omit)
g) Attachment 5-1: Laboratory Participation in Proficiency Testing/Measurement Audit Overview
h) Attachment 5-2: List of Laboratories Participating in Inter-Laboratory Comparisons (Optional if not participating in inter-laboratory comparisons)
i) Exhibit 6: Management System Audit Checklist (to be completed upon initial application)
Appendix 7: Laboratory English Proficiency Scope Table (to be filled out when English certificate attachments are required to be disclosed)
Appendix 7-1: Overview of Critical Laboratory Locations (English)
Appendix 7-2: List of Authorized Signatories for Laboratory Approval Applications (English)
Appendix 7-3: Scope of Accredited Testing Capabilities (English)
n) Appendix 7-8: Criteria Overview Table (English)
3. Application Attachment: Acknowledged Contract
4. Application materials submitted with the application form:
a) Proof of legal status of the laboratory, including the business license of the legal entity, and authorization documents from the legal entity or its legal representative (if the laboratory is not an independent legal entity)
b) Current valid quality manual (if available) and procedure documents (or any other name)
c) Documentation of the most recent complete internal audit and management review conducted by the laboratory (submitted at the time of initial application)
d) Laboratory Floor Plan
e) Description of the relationship when the laboratory is part of a legal entity
f) Verification of the current validity of the standards/methods applied for recognition (submit verification report)
g) Typical Project Inspection Reports/Calibration (Reference Measurement) Certificates/Authentication Documents and their Uncertainty Assessment Reports