



.jpg)
Summary
An electrodialysis unit is a water treatment technology that utilizes the selective permeability of ion exchange membranes to selectively direct the migration of ions in water under the influence of a direct current electric field, separating positive and negative ions in the solution. It is characterized by its low energy consumption, high water production, high desalination rate, and strong stability. Due to these features, it is used in various applications such as electronics, chemical industry, food processing, hard water softening, and seawater desalination.
Working Principle
The key component of an electrodialyzer is the ion exchange membrane, which comes in two types: cation exchange membrane (positive membrane) and anion exchange membrane (negative membrane). The positive membrane allows cations in the solution to pass through while blocking anions, whereas the negative membrane permits anions to pass through while blocking cations. Under the influence of an external electric field, ions in water move selectively in a directional manner, resulting in a reduction of ion concentration in one part of the water and an increase in another, thereby achieving desalination.
Key Technical Parameters
Turbidity < 1 mg/L
Total Iron Content < 0.3 mg/L
Total Manganese Content < 0.1 mg/L
Water temperature: 5-40°C
Chroma < 20℃
Oxygen Demand < 2mg/L (Oxygen based, Potassium permanganate method)
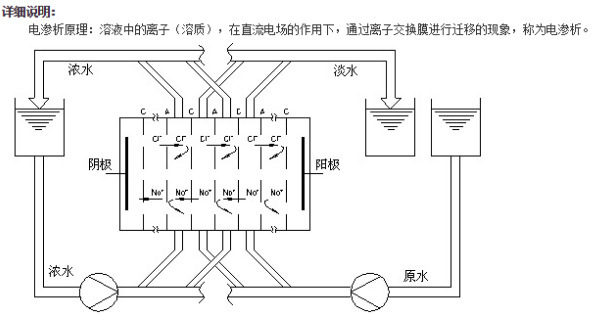
Electrodialysis units utilize the electroosmotic phenomenon to cause ion migration in the electrolyte solution within the compartments of the unit. This results in the separation of positive and negative ions, which pass through cation and anion exchange membranes to achieve the goals of dilution and concentration. Taking sodium chloride as an example, when a water solution containing sodium chloride passes through the compartments of an electrodialysis unit, which are lined with alternating cation and anion exchange membranes and freshwater and brine separators, ion migration occurs under the influence of a direct current field. Chloride ions migrate towards the anode, while sodium ions migrate towards the cathode, as shown in the diagram. Due to the selective permeability of the ion exchange membranes, the anion exchange membrane only allows anions to pass through, and the cation exchange membrane only allows cations to pass through. Therefore, cations in the dilution compartment migrate towards the cathode, passing through the anion membrane into the concentration compartment, while anions in the dilution compartment migrate towards the anode, passing through the cation membrane into the concentration compartment. In the concentration compartment, the ions also migrate towards the cathode and anode under the influence of the direct current field, but are blocked by the ion exchange membranes on both sides of the compartment, remaining within the concentration compartment. This causes the concentration of the solution in the concentration compartment to gradually increase, while the salt content in the dilution compartment gradually decreases. The electrodialysis unit is composed of alternating layers of dilution and concentration compartments, with half being dilution compartments and the other half being concentration compartments. Water from each compartment is collected through specially designed channels at the edges of the separators, forming a system of concentrated and diluted water to achieve desalination.
Electro dialysis (ED) unit composition
Electrodialysis equipment consists of a power supply converting AC to DC, an electrodialyzer, a cleaning device, and more. The main components of the electrodialyzer include an anion exchange membrane (abbreviated as anion membrane), a cation exchange membrane (abbreviated as cation membrane), diaphragms, electrodes, and clamping devices. The compartments formed by the diaphragms serve as channels for the liquid flow. By alternating the arrangement of the anion membrane, cation membrane, and diaphragms separating brine and freshwater, adding a pair of electrodes, and then clamping them together, an electrodialyzer is formed.
The electro dialysis membrane separator driven by an electric pump should operate normally under the following incoming water quality conditions:
Water Temperature: 5-40℃
b. Manganese Dioxide Index < 3 mg/L
c. Free Chlorine <0.2 mg/L
d. Iron <0.3 mg/L
e. Manganese <0.1 mg/L
f. Turbidity 1.5mm to 2.0mm divider <3 degrees
0.5mm to 0.9mm Separator <1 degree


















































