Technical Specifications
【CDE Platform Registration No.】: F20180000895 (Intravenous), F201800008146 (Oral), F20209990335 (Oral) Status A, F20210000552 (Intravenous) Status I
【USA DMF Registration Number】: 033403
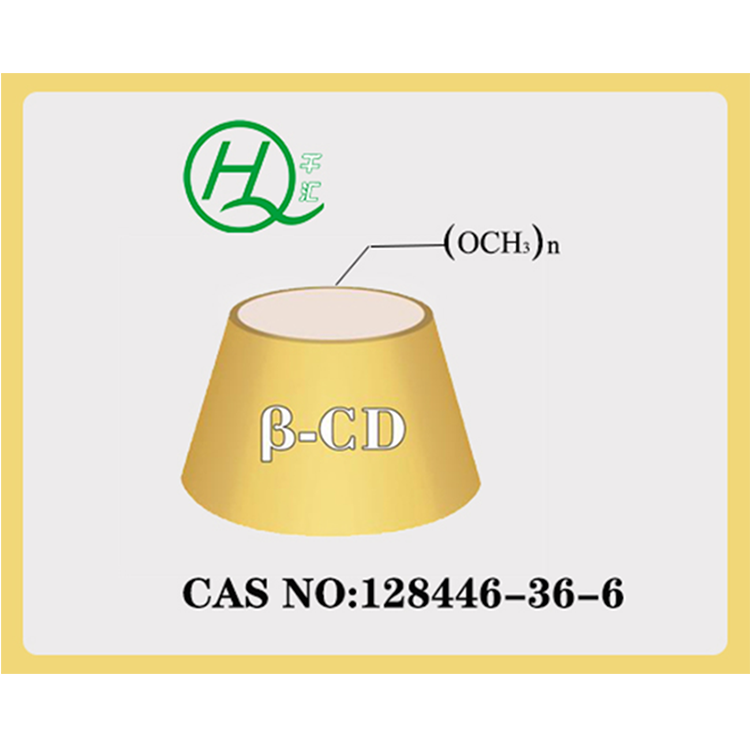
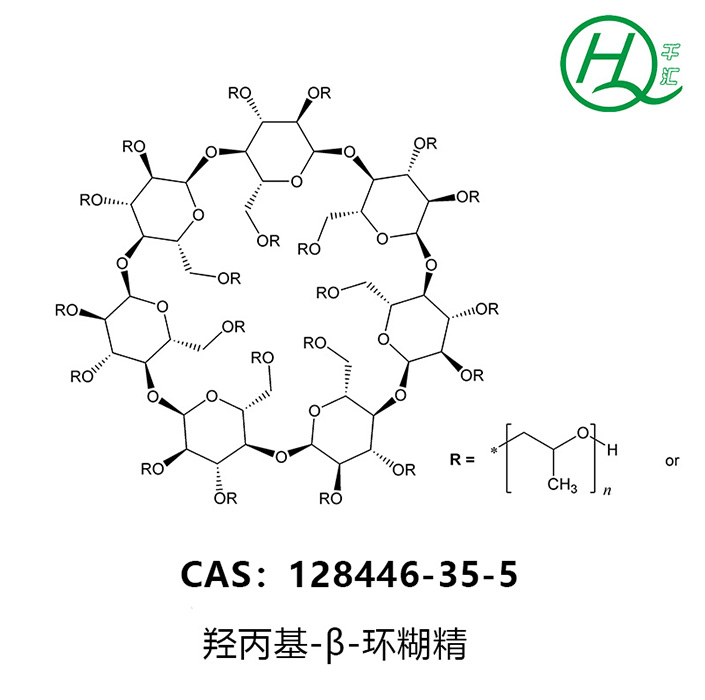
HYDROXYPROPYL-BETA-CYCLODEXTRIN
【CAS NO】:128446-35-5(94035-02-6)
【Molecular Formula】: C42H70-nO35(C3H7O)n, where n represents the degree of substitution
【Molecular Weight】: 1134 + 58n
【Level】:Injections and Oral
【Quality Standards】: USP-NF 2022, EP 10.0, ChP 2020, Corporate Standard
【Packaging】: 20KG/box, 20KG/drum, 10KG/drum, 1KG/bag, 0.5KG/bag
Application
In the MED field
Hydroxypropyl-β-cyclodextrin can solve the solubility issues of insoluble or poorly soluble substances (including plant extracts).
Through molecular encapsulation, enhance bio-solubility, control release rate, improve bioavailability, enhance stability, and reduce toxic and side effects.
In the food industry
Hydroxypropyl-β-cyclodextrin enhances the stability and longevity of nutritional molecules, can mask or correct the unpleasant odors and tastes of food nutritional molecules, improves production processes and product quality, and extends the shelf life of the product.
In the cosmetics sector
Used as stabilizers, emulsifiers, deodorizers, and more in cosmetic ingredients, it reduces the irritation of organic molecules in cosmetics to the skin and mucous membranes, enhances the stability of active ingredients, prevents the volatilization and oxidation of nutrients; increases the water solubility of insoluble fragrances and spices; and maintains a sustained and long-lasting fragrance release.