Guangzhou Microbiology Research is a professional clean environment testing organization, center, and company. We offer third-party testing, debugging, and consulting services for various clean environments. In accordance with clean environment testing standards, we use professional testing methods to issue qualified clean environment test reports. Additionally, our clean environment testing fees and prices are within a reasonable range.
Our center has passed the CNAS certification and CMA metrology certification. The test reports accurately reflect the actual conditions of the factory and can serve as a basis for third-party impartial evaluation. They are also applicable for QS and GMP certifications.
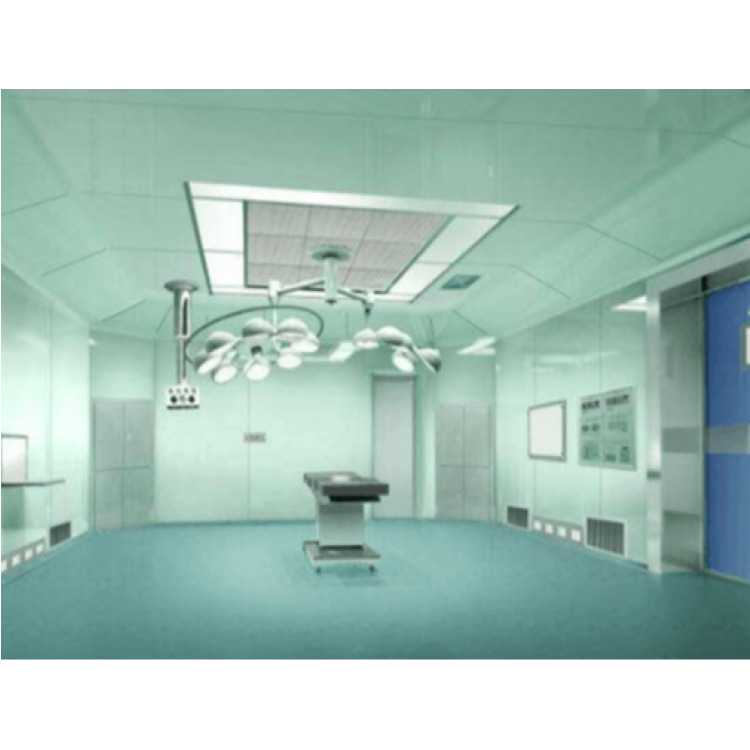
As an essential public service facility, the quality of its environment directly impacts its effectiveness. Standards such as 50333-2013 for Clean Operating Room Building Technology, WS 310.1-2016 for Sterile Supply Center – Part 1: Management Standards, GB15982-2012 for Sterilization Hygiene Standards, and WS 436-2013 for Operation Management of Secondary Water Supply, all provide specific regulations for indicators such as clean operating rooms, secondary water supply, indoor air quality, surface disinfection quality, and sterilization effectiveness of medical devices. We can provide scientific and authoritative inspection reports based on these standards for the acceptance, normal operation, and daily monitoring of related clean engineering projects.