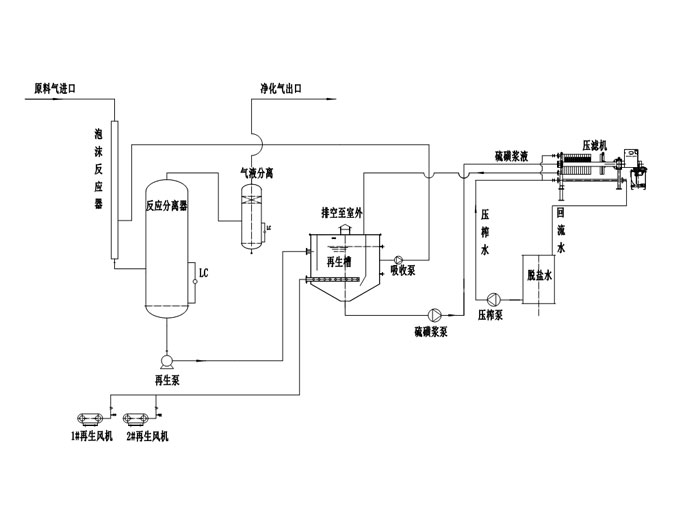
Basic Principle of Wet Flue Gas Desulfurization with Chelating Iron
Desulfurization Principle
Total Absorption Oxidation Reaction Equation
H2S(g)+2Fe3+(L)→2H+(L)+S↓+2Fe2+(L)
2. Recycling Principle
To restore the solution's ability to absorb hydrogen sulfide, the solution is regenerated, with the regeneration process primarily involving the oxidation and precipitation of sulfur.
1/2O2(g)+H2O(L)+2Fe2+(L)→2OH-(L)+2Fe3+(L)
Section II: Technical Features
High desulfurization efficiency.
(2) The system boasts strong anti-vibration capabilities.
(3) Low operating costs.
(4) The process has high selectivity and no by-products.
(5) High sulfur capacity.
(9) No wastewater discharge, no hazardous waste generation.
Section 3: Application Range: Hydrogen sulfide removal from biogas, associated petroleum gas, and industrial vent gas, with gas flow rate less than 100,000 Nm3/d; no limit on the hydrogen sulfide content in the feed gas; sulfur removal accuracy less than 10 PPm.