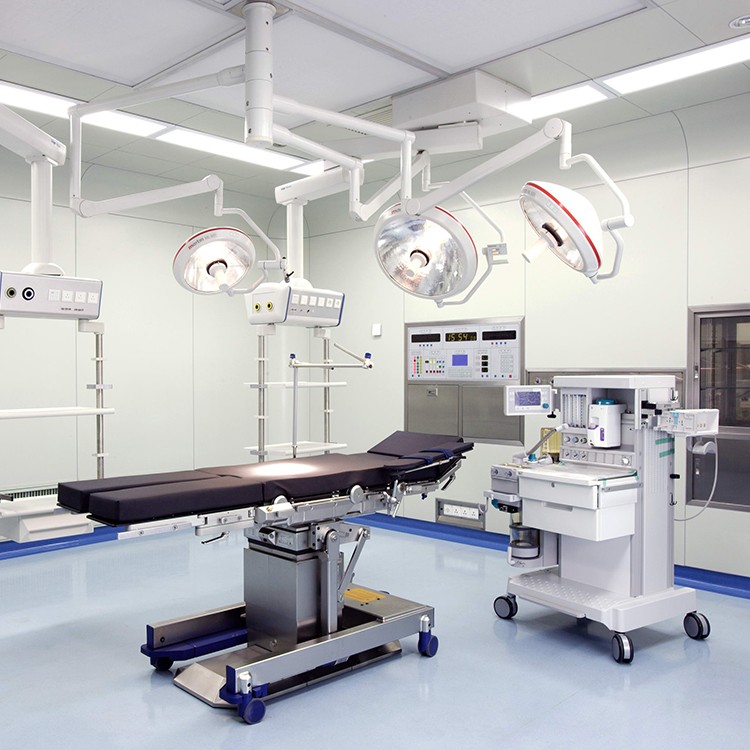
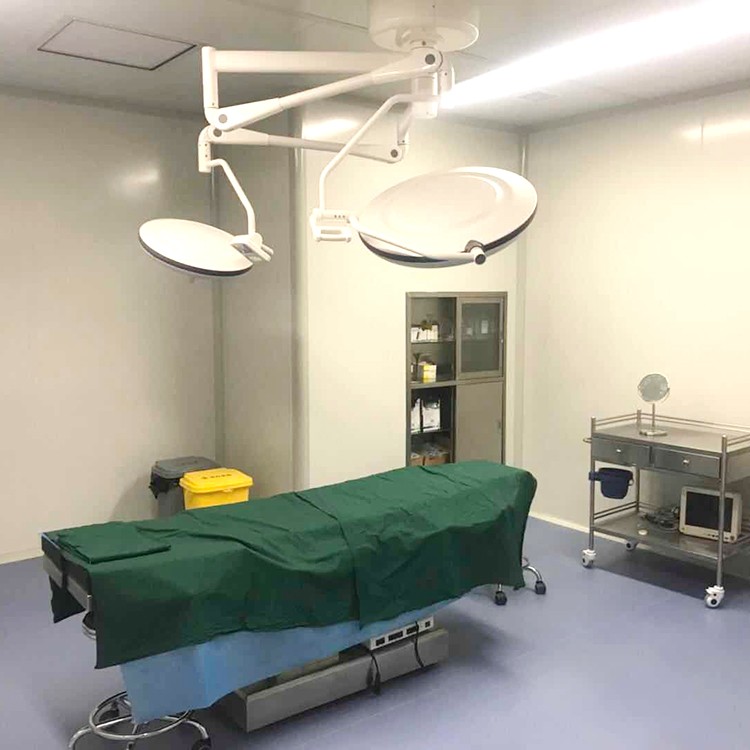
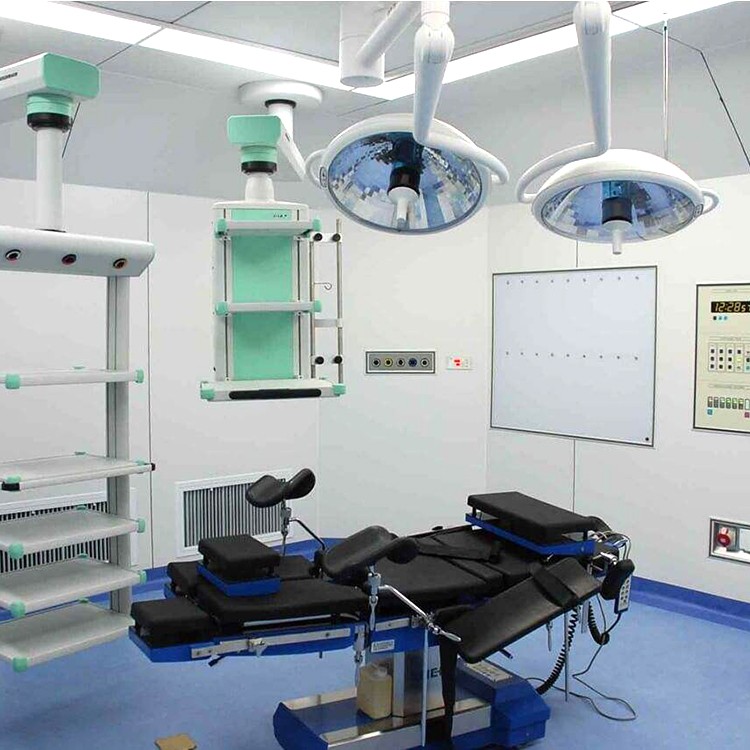
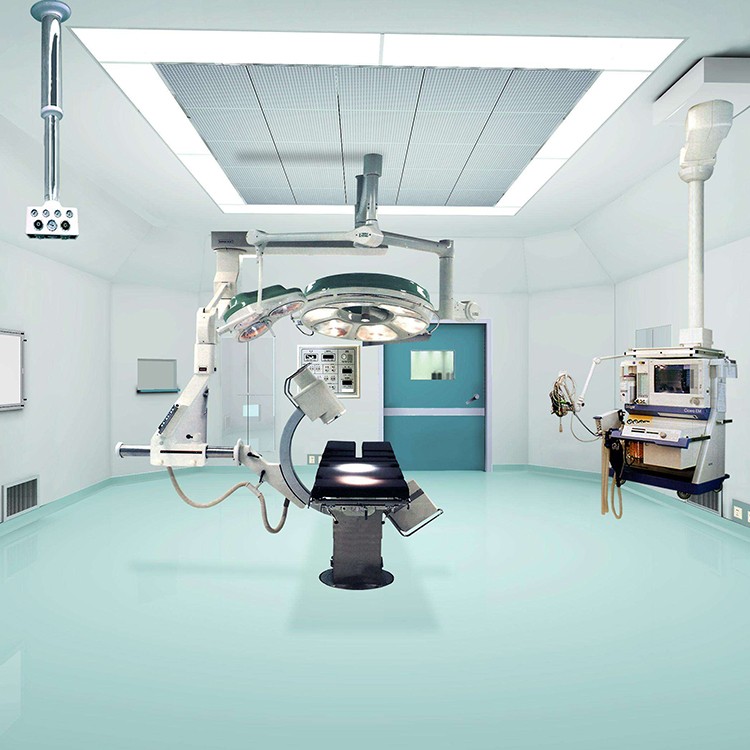
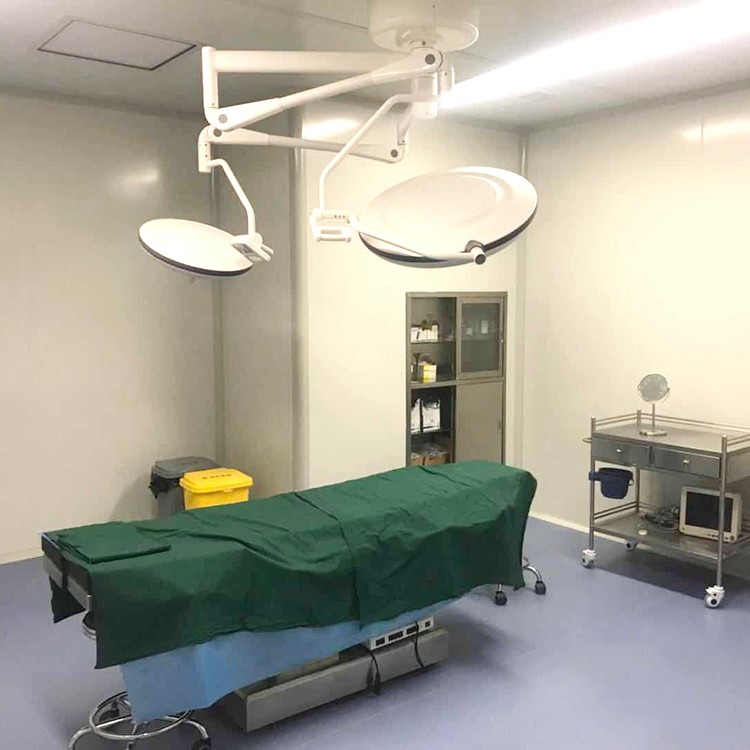
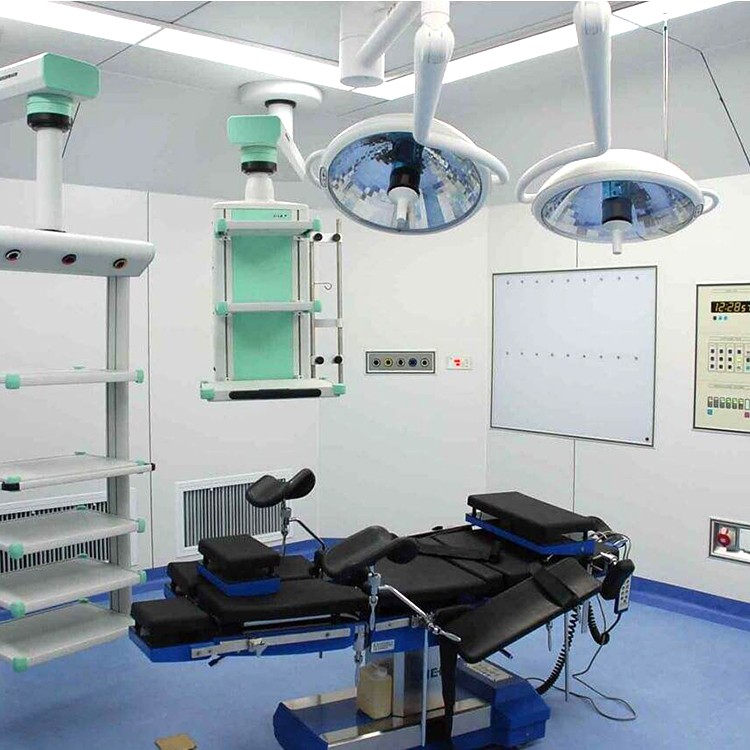
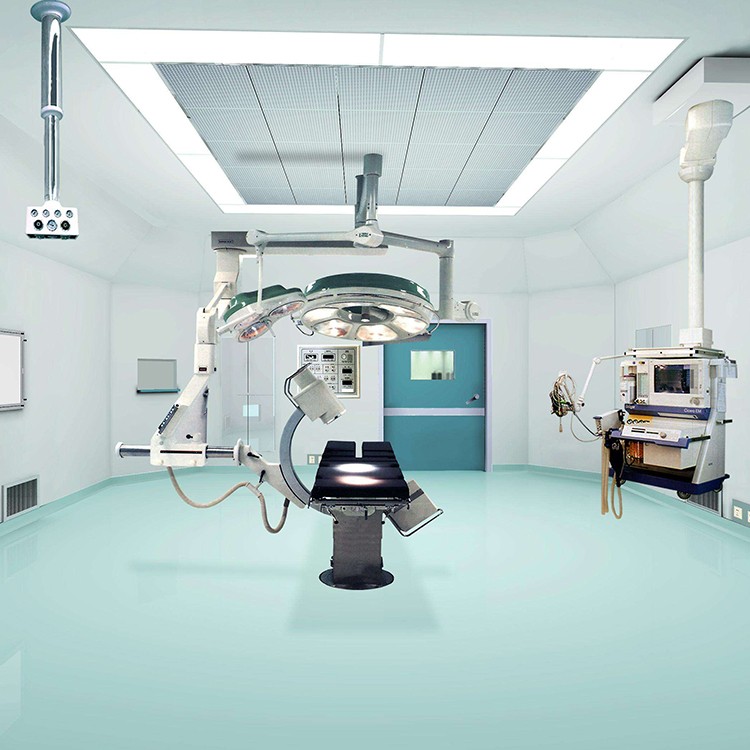
Precautions for using laminar flow operating room
1. Strengthen the training and management of operating room nurses
To effectively manage the purification project in the operating room, it is necessary to have a sound management system and quality control standards, conduct strict inspections and supervision, promptly identify and address problems, establish ideological awareness among management personnel, conscientiously implement rules and regulations management, business process management, and grasp various tasks.
The floor, walls, doors, and windows of the operating room should be kept clean every day.
The operating room decoration company adopts laminar flow operating room, and strengthening the concept and standardized management are still the fundamental and requirements of the medical process. The key to operating room work lies in the close integration of staff and cleanliness of the operating room. Before the surgery every morning, wipe the decoration of the operating room with a chlorine solution containing 500 milligrams per liter. ”After surgery, tables, starlight lamps, doors and windows in the operating room need to be wiped with 84 milligrams of detergent.
3. Ultraviolet radiation must be used for no less than 1 hour per week and thoroughly cleaned.
The purification project of the operating room should be entered by staff, and the operating room should be strictly carried out in accordance with regulations. The sewage treatment after operation should be carried out, and other instruments and auxiliary materials should be placed in fixed positions and treated by dedicated personnel around. After being subjected to high pressure, open the boiler door in the clean area, remove (i.e. no viable bacteria) items, and place them directly into the room.
4. Strictly control (i.e. no live bacteria) containers and steam once a week; Regularly change the agent for soaked items.
Items that require soaking should be regularly treated with special structures: capsules, flagella, and pili should be cultured simultaneously and a list should be kept for future inspection to determine if the measures (indicating solutions to problems) are appropriate; Surgical instrument automation, with instruction cards placed on each packaging. In order to indicate the date and product name on the packaging. If surgical supplies or infectious surgical instruments are contaminated, different instruments and items should be selected according to different situations, such as soaking, dressings, etc.