Phosphate ore, as a crucial raw material in the chemical industry and agricultural production, plays a significant role. Although China is rich in phosphate ore resources, most of them are low-grade, difficult-to-select ores. Therefore, the beneficiation and comprehensive utilization of low-grade phosphate ore are of great importance for promoting the sustainable development of the phosphate industry.
In China, phosphate rocks are categorized into three main types based on their geological formation and mode of occurrence: magmatic rock-type apatite, sedimentary rock-type phosphate rock, and sedimentary metamorphic rock-type phosphorite. Among them, sedimentary phosphate rock deposits are the primary phosphate resources in China, accounting for over 80% of the country's total phosphate reserves. Extensive research has been conducted on the beneficiation of phosphate ore, covering both process flow and reagent research advancements. Liu Zhichao et al. [3] conducted flotation experiments on a phosphorus-iron symbiotic ore, using BK420-C as a collector, achieving a flotation concentrate with a P2O5 grade of 35.01% and a recovery rate of 52.81% after a single roughing, one scavenging, and three cleaning operations. Guo Wendao et al. [4] conducted a study on the flotation behavior of phosphorus in a phosphorus-containing iron ore, obtaining a phosphorus concentrate with a P2O5 grade of 30.74% and a recovery rate of 72.94% after a single roughing, three cleaning, and two scavenging operations under collector dosage of 600 g/t and water glass dosage of 400 g/t. Nan Nan et al. [5] focused on the beneficiation of pyrite concentrate from Baiquan, using a collector consisting of oxidized paraffin soap + fatty acid + MES, and achieved a phosphorus concentrate with a P2O5 grade of 33% and a recovery rate of 85% through a single roughing, three cleaning, one scavenging, and middlings return closed-circuit flotation process. Nie Yimiao et al. [6] in their phosphorus recovery tests, obtained a phosphorus concentrate with a grade of 32.74% and a recovery rate of 86.11% under conditions of -0.074 mm grind size at 52.1%, pH of 8.5-9, water glass dosage of 800 g/t, AW-01 dosage of 800 g/t, pulp concentration of 30%, roughing time of 3 minutes, water glass dosage of 50 g/t during cleaning, AW-01 dosage of 100 g/t, and pulp concentration of 25%, using a single roughing and three cleaning flotation process. Qin Yufang [7] and others, based on ore property research, employed sodium carbonate as a pulp pH adjuster, water glass as a gangue mineral inhibitor, and sodium laurate as a phosphorus mineral collector, achieving a phosphorus concentrate with a P2O5 grade of 30.21% and a recovery rate of 70.81% through a single roughing and two cleaning closed-circuit test process.
In summary, the exploration of phosphate ore beneficiation processes focuses on grinding fineness, pulp pH, types of collectors, and the number of concentration and cleaning cycles. The Liaoning region is predominantly composed of metamorphic rock-type apatite mines, which have low apatite content. Although the ore is relatively easy to process, the beneficiation ratio is high, resulting in high concentrate costs. This article, targeting iron-phosphate ore in a certain area of Liaoning, explores the types, dosages, and interactions of reagents based on the study of ore properties, providing data references for the field.
1. Raw ore characteristics
1.1 Preparation of Test Ore Sample
The test ore samples were provided by a mining company in Liaoning Province. The ore samples were processed through three stages of crushing. The coarse crushing was carried out using a PE-150×200 jaw crusher to reduce the ore to less than 50 mm; the medium crushing used an XPC-60×200 jaw crusher to break the minerals down to less than 12 mm. The crushed ore samples were mixed and subdivided; a portion was fed into a roller crusher for further crushing, while the remainder was retained as backup samples. The fine crushing was performed using an XPS-Φ250×150 roller screen to reduce the ore to less than 2 mm, and a 2 mm sieve was used for screening. The samples below 2 mm were mixed and subdivided; 100 g of ore samples were taken for chemical full element analysis and chemical multi-element analysis, and the remaining ore samples were bagged in increments of 1,000 g for subsequent flotation tests.
1.2 Sample Nature
1.2.1 Comprehensive Chemical Elemental Analysis
To investigate the chemical composition of the ore samples, qualitative and semi-quantitative full-element chemical analysis was conducted. The analysis employed X-ray fluorescence (XRF) methodology. The test results are presented in Table 1.
The chemical full element analysis results in Table 1 indicate that the main valuable components of the ore are phosphorus and iron, with titanium potentially recoverable, and the main gangue mineral components are silicate-aluminate and alkaline calcium-magnesium compounds. Consequently, the next phase of chemical analysis will focus on the potentially usable valuable mineral components such as P2O5, Fe2O3, and TiO2, the primary gangue components including SiO2, Al2O3, CaO, MgO, and the contents of other elements like Na2O, K2O, and SO3.
1.2.2 Chemical Multi-element Analysis
Based on the X-ray fluorescence analysis results in Table 1, except for the low content of MnO, Cl, ZnO, CuO, NiO, BaO, and SrO, quantitative analysis was conducted on the other chemical elements, with the results displayed in Table 2.
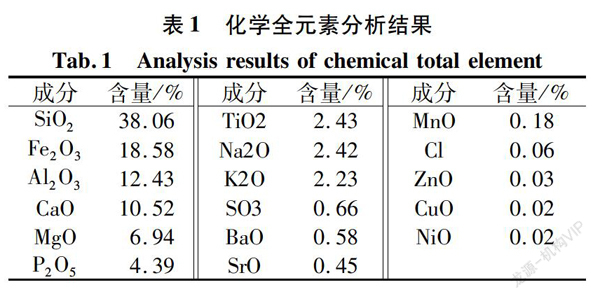
Based on the chemical multi-element analysis results in Table 2, it is further determined that the main useful components in the ore are phosphorus and iron. These can be comprehensively recovered as apatite and iron ore during the flotation process. Titanium content is low, and it is not considered for separate recovery. The gangue minerals are primarily quartz.
1.2.3 Composition of Ore
The valuable minerals in the ore mainly include apatite, magnetite, and rutile, followed by pyrite, chalcopyrite, and pyrrhotite. The gangue minerals consist of diopside, potassium feldspar, plagioclase, biotite, calcite, dolomite, talc, quartz, sphene, and rutile. The mineral quantification of the main ore minerals reveals that the relative content of apatite is 4.28%, magnetite is 12.50%, and rutile is 3.50%. The remainder is primarily composed of pyroxene and feldspar, with some biotite, a small amount of quartz, carbonate minerals, talc, and metallic sulfides. The relative content of minerals in the ore is shown in Table 3.
2 Flotation Test
2.1 Absorbent Agent Variety Test
Phosphorite flotation primarily employs fatty acid collectors, with representative fatty acid collectors including oleic acid, sodium oleate, and paraffin wax soap [8-11]. In this trial, in addition to using the aforementioned common collectors, the research group's homemade collectors JZQ-F7 and JZQ-F2, both fatty acid collectors, will also be examined.
During the flotation test, a roughing and two scavenging flotation process was adopted. The pulp pH was 8.5 to 9, with a flotation temperature of 25 to 30 ℃. The collectors used were oleic acid, sodium oleate, paraffin wax soap, JZQ-F7, and JZQ-F2. The dosage of inhibitor water glass was 1,800 g/t, with the rougher collector dosage at 1,000 g/t, the first scavenger dosage at 400 g/t, and the second scavenger dosage at 200 g/t. The test results are shown in Figure 1.
Figure 1's flotation test results of different collectors reveal that, based on the analysis of the P2O5 grade in the concentrate, while oleic acid can raise the P2O5 grade in the concentrate to above 12%, under the influence of the other four collectors, the P2O5 grade in the concentrate remains around 8% to 9%, indicating a relatively similar selectivity among collectors. In terms of concentrate yield and recovery rate, the concentrate recovery rate was 30.90% under the action of oleic acid, 19.19% under sodium oleate, 15.42% under JZQ-F2, and 91.82% and 91.76% respectively under JZQ-F7 and paraffin wax soap. The data suggest that although sodium oleate can achieve a higher concentrate grade, the recovery rate is lower. However, the concentrate grade can be improved through subsequent purification, so the concentrate recovery rate should be prioritized in the initial concentration. Therefore, under the above flotation conditions, JZQ-F7 and paraffin wax soap can achieve relatively good test indicators. To simplify the experiment, subsequent tests will use JZQ-F7 as the collector to investigate the relationship between collector dosage and interaction with other chemicals.
2.2 Examination of the Interaction Between Sodium Carbonate, Silicate Gel, and JZQ-F7
Based on literature research, the flotation pH for apatite typically ranges between 10 to 11 to achieve good flotation test results [12-14]. Therefore, the study investigated the flotation effect of sodium carbonate, a pH adjuster, as well as its interaction with inhibitors such as water glass and collector JZQ-F7. To simplify the experiment, a single roughing test was selected, with the test process depicted in Figure 2. The results are shown in Figure 3 (T represents sodium carbonate, S represents water glass).
Figure 3 flotation test data indicates that, based on the analysis of the concentrate P2O5 grade and recovery rate, the reagent combination of sodium carbonate, water glass, and JZQ-F7 achieves better flotation test results compared to other reagent combinations. The concentrate P2O5 grade reaches 12.19%, with a P2O5 recovery rate of 95.64%. The next experiment will discuss the relationship between the dosages of the three reagents.
2.3 Orthogonal Experimentation on Dosage of Three Medicines
Based on the 2.2 flotation test data, it is evident that a combination of sodium carbonate, water glass, and JZQ-F7 reagents achieves favorable flotation test indicators. An optimization test was conducted on the dosage of the three reagents, utilizing an orthogonal design method. The type of reagents was considered as the factor affecting the flotation test, with the dosage of each reagent as the level of the factor, and the P2O5 grade and recovery rate of the flotation concentrate as the indicators of the test results. The test process is shown in Figure 4, the setting of factor levels is listed in Table 4, and the test results are presented in Table 5.
During the test, sodium carbonate and water glass were added to the rough selection process in full. The collector JZQ-F7 was added in proportion to both the rough selection and the two sweep selections. When the collector dosage was 1,200 g/t, 700 g/t was added to the rough selection, 300 g/t to the first sweep, and 200 g/t to the second sweep; when the collector dosage was 1,800 g/t, 1,000 g/t to the rough selection, 500 g/t to the first sweep, and 300 g/t to the second sweep; when the collector dosage was 2,400 g/t, 1,300 g/t to the rough selection, 800 g/t to the first sweep, and 300 g/t to the second sweep.
Based on the orthogonal test data from Tables 5 and 6, it is evident that the rank of R grade is C > B > A, and the rank of recovery rate is C > B > A, indicating that the primary factors affecting the test indicators are C > B > A. Higher concentrations of the concentrate recovery rate and grade are preferable. Therefore, higher values for the I, II, and III parameters are desired. For grade, level A2B3C1 is optimal, while for recovery rate, level A3B1C3 is superior. At the A2B3C1 level, using 1,500 g/t of sodium carbonate, 2,400 g/t of water glass, and 1,200 g/t of collector yields a concentrate P2O5 grade of 12.19% and a recovery rate of 95.64%. At the A3B1C3 level, using 2,000 g/t of sodium carbonate, 1,200 g/t of water glass, and 2,400 g/t of collector results in a concentrate P2O5 grade of 8.09% and a recovery rate of 97.26%. Comparing the two sets of indicators, the P2O5 grade of the concentrate at the A2B3C1 level is significantly higher than that at the A3B1C3 level, by 4.10 percentage points. The P2O5 recovery rate at the A2B3C1 level is slightly lower than that at the A3B1C3 level, by 1.62 percentage points, and the A3B1C3 level achieved a higher recovery rate by substantially increasing the collector dosage, which also increased the yield of the concentrate. To ensure the P2O5 grade of the rough concentrate for subsequent flotation, it is necessary to control the yield of the rough concentrate and avoid excessive collector dosage. Therefore, the A2B3C1 level is selected, which is 1,500 g/t of sodium carbonate, 2,400 g/t of water glass, and 1,200 g/t of collector.
2.4 Selective Test Runs
To investigate the impact of the number of concentration cycles on the concentrate product, four concentration tests were designed, with the number of concentration cycles as the variable. A single rough concentration was used, with different numbers of concentration cycles in the test process. The rough concentration test conditions were the same, with 1,500 g/t of sodium carbonate, 2,400 g/t of water glass, and 1,200 g/t of JZQ-F7. The test results are shown in Figure 5.
Figure 5 shows that as the number of selected times increases, the grade of the concentrate P2O5 gradually rises, while the recovery rate of the concentrate P2O5 gradually decreases, which conforms to the general flotation rule. When the selected times reach four, the P2O5 grade of the concentrate reaches 31.42%, meeting the standard for phosphorus concentrate products. Therefore, the design of the closed-loop test for the number of selected times is set at four.
2.5 Flotation Closed-Circuit Test
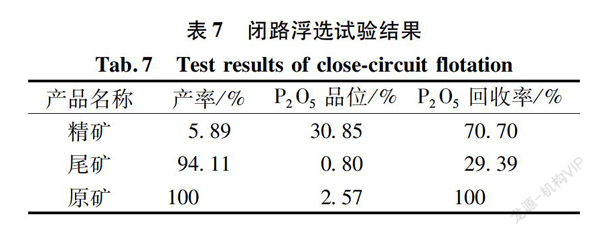
To assess the impact of mineral products entering the beneficiation system on flotation indices, a closed-circuit test was conducted. Typically, the grade of the concentrate product in the closed-circuit test is reduced, while the recovery rate is improved. The closed-circuit test was designed based on the rough and clean flotation conditions, with the test process depicted in Figure 6 and the results shown in Table 7.
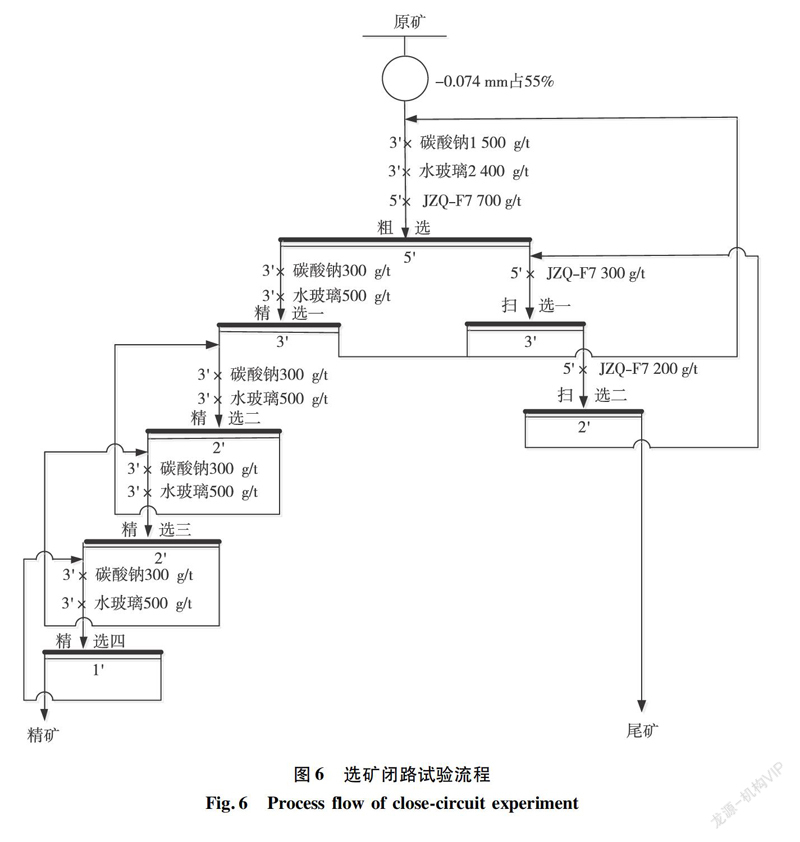
Table 7 indicates that through a closed-circuit flotation process test involving one roughing, two scavenging, and four cleaning stages, followed by the return of middlings, a phosphorus concentrate with a P2O5 grade of 30.85% and a recovery rate of 70.70% can be achieved.
3. Conclusion
1) Analysis of the ore sample indicates that the main useful components are phosphorus and iron, with potential recoverable components including titanium. The main gangue mineral composition consists of silicate-aluminates and alkaline calcium magnesium compounds. The potentially usable mineral components include P2O5, Fe2O3, TiO2; the primary gangue components are SiO2, Al2O3, CaO, MgO.
2) Flotation test studies indicate that when the grinding fineness reaches 55% of -200 mesh, using crude soda as a pH adjuster at a rate of 1,500 g/t, sodium silicate as an inhibitor at a rate of 2,400 g/t, and JZQ-F7 as a collector at a rate of 700 g/t, four consecutive concentration processes yield an open-circuit indicator of phosphorus concentrate with a P2O5 grade of 31.42% and a recovery rate of 68.46%, as well as a closed-circuit indicator of phosphorus concentrate with a P2O5 grade of 30.85% and a recovery rate of 70.70%.
Research in the field of process mineralogy reveals that, in the ore samples, in addition to phosphorus, iron is also a valuable mineral element, primarily existing in magnetite form. There has been considerable research into separately recovering phosphorus and iron from phosphorus-iron ores. Magnetic separation can be employed to recover magnetite from the flotation tailings.