The selection of reverse osmosis membrane elements should be considered comprehensively, taking into account factors such as the raw water type and salinity, water production and quality, system recovery rate, and desalination rate requirements; for reverse osmosis membrane elements, the salt content (TDS) they can withstand, brine osmotic pressure, and pressure should also be considered.

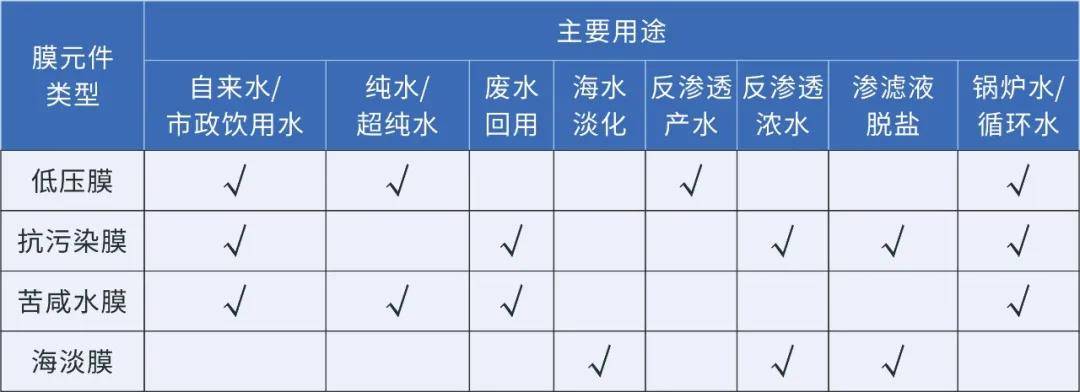
Choose reverse osmosis membranes based on application scenarios.
Low-pressure membranes are commonly used in industrial pure water, boiler feed water, tap water purification desalination, and as secondary treatment for reverse osmosis product water. Even under lower operating pressures, they still maintain high desalination rates.
The brackish water membrane is primarily used for desalination treatment of surface water, municipal water, and industrial wastewater, as well as for recycled water reuse processing. It effectively reduces the frequency of chemical cleaning, lowers operational costs, and boasts high desalination rates, high water yields, and high chemical durability.
Anti-pollution membranes are commonly used in highly polluted water sources, such as severely polluted industrial wastewater, offering superior anti-pollution capabilities for organic matter and microorganisms in the incoming water. They also possess high chemical durability, effectively addressing the issue of membrane element contamination.
Desalination membranes are primarily used in seawater desalination, reverse osmosis brine treatment, leachate, and other highly contaminated water sources.
II. Different Total Dissolved Solids (TDS) lead to varying brine osmotic pressure and pressure.
Due to varying Total Dissolved Solids (TDS), osmotic pressure differences are determined, which also lead to different operating pressures of the system. Osmotic pressure is typically calculated using the following formula:
π=RT∑Ci
R is a constant, usually taken as 0.082 atm·L/mol·K.
∑Ci is the total concentration of each ion, in mol/L.
T stands for thermodynamic temperature, K
What is the osmotic pressure of a 2000ppm sodium chloride solution at 25°C?
∑Ci = C(Na+) + C(Cl-) = 0.0684 mol/L
π=RT∑Ci=0.082X(273+25)X0.0684=1.6714atm=16881KPa
The reverse osmosis process can only be achieved when the operating pressure exceeds the concentrate osmotic pressure, meaning the feedwater pressure must be greater than 168KPa. When selecting membrane elements, we need to estimate the concentrate osmotic pressure and pressure based on the raw water ion concentration or salt content. It is essential to ensure that the pressure is greater than the concentrate osmotic pressure while not exceeding the membrane's pressure limit. Excessive pressure can lead to damage to the membrane elements. On the other hand, when considering the high-pressure pump head, it is also necessary to select an appropriate head based on the pressure.
Additionally, the system's recovery rate affects the thick water TDS, which in turn has an impact on pressure.
For example, with a raw water concentration of 5000ppm (based on NaCl) and a recovery rate of 75%, the concentrated water TDS is equivalent to a 4x concentration, which means the concentrated water TDS is 20,000ppm. At this point, the osmotic pressure is approximately 16.8 bar. If the operating pressure of a reverse osmosis membrane is 41 bar, this membrane can be chosen under such conditions. However, if the raw water TDS is 15,000ppm and the recovery rate is still 75%, the concentrated water TDS would be 60,000ppm. The osmotic pressure would then exceed 41 bar, surpassing the pressure limit of the membrane element. In this case, the reverse osmosis membrane should not be used, and a higher-pressure-grade seawater desalination membrane should be chosen instead.
Choose reverse osmosis membranes based on the total dissolved solids (TDS) content of the incoming water.
For different reverse osmosis membranes, there are variations in the salt content they can withstand during operation. Therefore, it is essential to select the appropriate membrane element based on the actual salt content of the incoming water (i.e., TDS).
Water TDS ≤ 1000ppm, such as tap water, surface water, and reverse osmosis product water, typically uses low-pressure membranes.
Inlet TDS ≤ 8000ppm, for saline wastewater such as municipal and industrial wastewater, generally use brackish water membranes or antifouling membranes. For water sources with severe organic and microbial contamination, antifouling membranes must be selected.
For water with TDS>8000ppm, such as brackish water with high salt content, reverse osmosis concentrate, leachate, seawater, etc., desalination membranes or high-pressure brackish water membranes are typically selected.
In summary,The selection of reverse osmosis membranes must take into account factors such as water quality of the produced water, energy consumption per ton of water, and the membrane's stability against pollution.





