I. Overview
Sponge iron deaerator is a device for oxygen removal, where water containing oxygen is fed into the deaerator and passes through a layer of sponge iron filter material. This specially designed sponge iron filter material has a vast specific surface area, enabling a complete reaction between the dissolved oxygen in the water and the iron.

Section 2: Working Principle
Room temperature filtration deoxygenation is a chemical deoxygenation process. It uses specially produced activated sponge iron (directly reduced iron) to remove dissolved oxygen from water. The sponge iron primarily consists of iron, with a loose and porous internal structure that provides a specific surface area up to 50,000 to 100,000 times greater than that of ordinary iron filings. This allows for a rapid and complete oxidation reaction between the oxygen in the water and the iron, stabilizing the dissolved oxygen level below 0.05 mg/L. The reaction equation is: 2Fe2+ + 2H2O + O2 → 2Fe(OH)2
4Fe(OH)2+ 2H2O +O2→4Fe(OH)3[1]
The reaction products Fe(OH)2 and Fe(OH)3 are non-soluble, flocculent precipitates. They are intercepted as the water flows through the remaining sponge iron particles. They can be easily cleaned by a stream of backwash with moderate strength (about 5 minutes).
The consumption of sponge iron is low, typically requiring a replenishment every 3-6 months, depending on the treated water volume and quality.
Deoxygenated water enriched with a small amount of iron ions, typically 0.2 - 0.5 mg/L, still meets the water quality standards for hot water boilers. However, for steam boilers or deoxygenation processes with strict requirements for Fe2+ in feedwater, an iron removal device can be added to eliminate Fe2+ from the water. The sponge iron used as a deoxygenation agent is made by bonding together Fe3O4 powder with particle sizes below -200 mesh, with particle sizes of about 2-5 mm. Due to the displacement reaction during the reduction process, it forms a certain number of voids, as well as voids from the bonding of fine particles, resulting in a high porosity and large specific surface area. Being directly reduced from primary ore, it is relatively pure, thus highly active and reacts vigorously with oxygen in the water to form Fe3O4, achieving the deoxygenation goal. At the same time, the utilization rate is also increased to over 90%. The results are highly satisfactory, with the oxygen content in the effluent reduced to below 0.05 mg/L, and only about 25g of treatment is required per cubic meter of water, ensuring high-quality, low-cost operation. Since water that hasn't been softened can cause the deoxygenation agent surface to钝ize, slowing down oxidation and affecting deoxygenation efficiency, it is recommended to soften the water first before deoxygenation. After some use, backwashing should be performed.

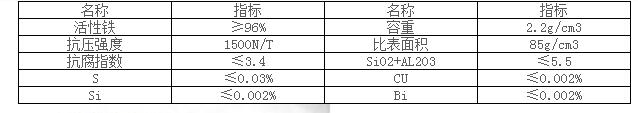
Section 3: Technical Parameters of Sponge Iron Filtration Material